13.3
Impact Factor
Theranostics 2022; 12(10):4513-4535. doi:10.7150/thno.72269 This issue Cite
Research Paper
Blockade of phosphotyrosine pathways suggesting SH2 superbinder as a novel therapy for pulmonary fibrosis
1. Department of Pathophysiology, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
2. Department of Medical Genetics, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
3. Department of Respiratory and Critical Care Medicine, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
4. Department of Pathology, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
5. Department of Oncology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
6. Department of Forensic Medicine, School of Basic Medicine, Nanjing Medical University, Nanjing, China
7. Department of Biochemistry, Schulich School of Medicine and Dentistry, Western University, London, Ontario, Canada
8. Key Laboratory of Respiratory Diseases, National Health Commission of China, Wuhan, China
# These authors contributed equally to this work.
Abstract
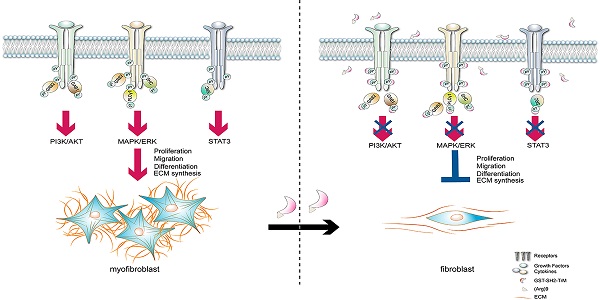
Background: Idiopathic pulmonary fibrosis (IPF) is a progressive and irreversible fibrotic disease with high mortality. Currently, pirfenidone and nintedanib are the only approved drugs for IPF by the U.S. Food and Drug Administration (FDA), but their efficacy is limited. The activation of multiple phosphotyrosine (pY) mediated signaling pathways underlying the pathological mechanism of IPF has been explored. A Src homology-2 (SH2) superbinder, which contains mutations of three amino acids (AAs) of natural SH2 domain has been shown to be able to block phosphotyrosine (pY) pathway. Therefore, we aimed to introduce SH2 superbinder into the treatment of IPF.
Methods: We analyzed the database of IPF patients and examined pY levels in lung tissues from IPF patients. In primary lung fibroblasts obtained from IPF patient as well as bleomycin (BLM) treated mice, the cell proliferation, migration and differentiation associated with pY were investigated and the anti-fibrotic effect of SH2 superbinder was also tested. In vivo, we further verified the safety and effectiveness of SH2 superbinder in multiple BLM mice models. We also compared the anti-fibrotic effect and side-effect of SH2 superbinder and nintedanib in vivo.
Results: The data showed that the cytokines and growth factors pathways which directly correlated to pY levels were significantly enriched in IPF. High pY levels were found to induce abnormal proliferation, migration and differentiation of lung fibroblasts. SH2 superbinder blocked pY-mediated signaling pathways and suppress pulmonary fibrosis by targeting high pY levels in fibroblasts. SH2 superbinder had better therapeutic effect and less side-effect compare to nintedanib in vivo.
Conclusions: SH2 superbinder had significant anti-fibrotic effects both in vitro and in vivo, which could be used as a promising therapy for IPF.
Keywords: IPF, phosphotyrosine (pY), fibroblast, SH2 superbinder, therapy
 Global reach, higher impact
Global reach, higher impact