13.3
Impact Factor
Theranostics 2022; 12(9):4399-4414. doi:10.7150/thno.72354 This issue Cite
Research Paper
Dysadherin awakens mechanical forces and promotes colorectal cancer progression
1. School of Life Sciences, Gwangju Institute of Science and Technology, Gwangju, 61005, Republic of Korea
2. Cell Logistics Research Center, Gwangju Institute of Science and Technology, Gwangju, 61005, Republic of Korea
3. Department of Hemato-oncology, Chonnam National University Medical School, Gwangju, 61469, Republic of Korea
4. Department of Pathology, Chonnam National University Medical School, Gwangju, 61469, Republic of Korea
*These authors contributed equally to this work.
# Current address: Institute for Basic Science, Center for Genome Engineering, 55, Expo-ro, Yuseong-gu, Daejeon, Korea, 34126
Abstract
Rationale: Dysadherin is a tumor-associated, membrane-embedded antigen found in multiple types of cancer cells, and associated with malignant behavior of cancer cells; however, the fundamental molecular mechanism by which dysadherin drives aggressive phenotypes of cancer is not yet fully determined.
Methods: To get a mechanistic insight, we explored the physiological relevance of dysadherin on intestinal tumorigenesis using dysadherin knockout mice and investigated its impact on clinicopathological features in patients with advanced colorectal cancer (CRC). Next, to discover the downstream signaling pathways of dysadherin, we applied bioinformatic analysis using gene expression data of CRC patient tumors and dysadherin knockout cancer cells. Additionally, comprehensive proteomic and molecular analyses were performed to identify dysadherin-interacting proteins and their functions.
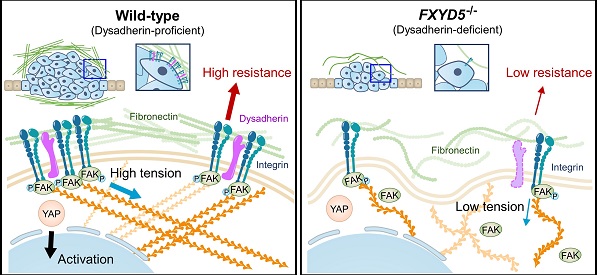
Results: Dysadherin deficiency suppressed intestinal tumorigenesis in both genetic and chemical mouse models. Moreover, increased dysadherin expression in cancer cells accounted for shorter survival in CRC patients. Comprehensive bioinformatics analyses suggested that the effect of dysadherin deletion is linked to a reduction in the extracellular matrix receptor signaling pathway. Mechanistically, the extracellular domain of dysadherin bound fibronectin and enhanced cancer cell adhesion to fibronectin, facilitating the activation of integrin-mediated mechanotransduction and leading to yes-associated protein 1 activation. Dysadherin-fibronectin interaction promoted cancer cell growth, survival, migration, and invasion, effects collectively mediated the protumor activity of dysadherin.
Conclusion: Our results highlight a novel function of dysadherin as a driver of mechanotransduction that stimulates CRC progression, providing a potential therapy strategy for CRC.
Keywords: Dysadherin, Mouse Model, Colorectal Cancer, Fibronectin, Yes-associated Protein 1
 Global reach, higher impact
Global reach, higher impact