13.3
Impact Factor
Theranostics 2022; 12(3):1440-1458. doi:10.7150/thno.58780 This issue Cite
Research Paper
Downregulation of low-density lipoprotein receptor mRNA in lymphatic endothelial cells impairs lymphatic function through changes in intracellular lipids
1. Department of Medicine, Faculty of Medicine, Université de Montréal, Montreal, Quebec, Canada.
2. Montreal Heart Institute, Research Center, Montreal, Quebec, Canada.
3. Department of Nutrition, Faculty of Medicine, Université de Montréal, Montreal, Quebec, Canada.
4. Montreal Heart Institute, Metabolomics platform, Montreal, Quebec, Canada.
5. Department of Medicine, Medical College of Wisconsin, Milwaukee, Wisconsin, USA.
6. Faculty of Pharmacy, Université de Montréal, Montreal, QC, Canada.
*Co-first authors.
Abstract
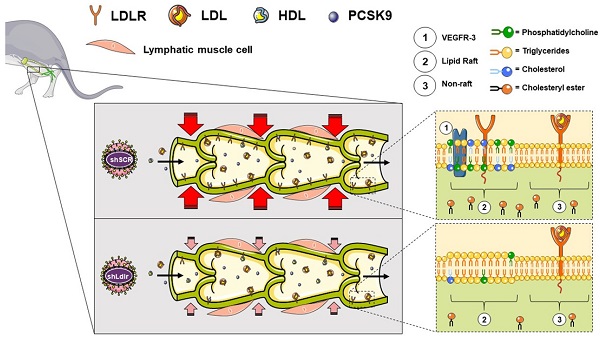
Rationale: Impairment in lymphatic transport is associated with the onset and progression of atherosclerosis in animal models. The downregulation of low-density-lipoprotein receptor (LDLR) expression, rather than increased circulating cholesterol level per se, is involved in early atherosclerosis-related lymphatic dysfunction. Enhancing lymphatic function in Ldlr-/- mice with a mutant form of VEGF-C (VEGF-C 152s), a selective VEGFR-3 agonist, successfully delayed atherosclerotic plaque onset when mice were subsequently fed a high-fat diet. However, the specific mechanisms by which LDLR protects against lymphatic function impairment is unknown.
Methods and results: We have thus injected wild-type and Pcsk9-/- mice with an adeno-associated virus type 1 expressing a shRNA for silencing Ldlr in vivo. We herein report that lymphatic contractility is reduced upon Ldlr dowregulation in wild-type mice only. Our in vitro experiments reveal that a decrease in LDLR expression at the mRNA level reduces the chromosome duplication phase and the protein expression of VEGFR-3, a membrane-bound key lymphatic marker. Furthermore, it also significantly reduced the levels of 18 lipid subclasses, including key constituents of lipid rafts as well as the transcription of several genes involved in cholesterol biosynthesis and cellular and metabolic processes. Exogenous PCSK9 only reduces lymphatic endothelial-LDLR at the protein level and does not affect lymphatic endothelial cell integrity. This puts forward that PCSK9 may act upon lymphatic muscle cells to mediate its effect on lymphatic contraction capacity in vivo.
Conclusion: Our results suggest that treatments that specifically palliate the down regulation of LDLR mRNA in lymphatic endothelial cells preserve the integrity of the lymphatic endothelium and sustain lymphatic function, a prerequisite player in atherosclerosis.
Keywords: Atherosclerosis, Lymphatic System, Lymphatic dysfunction, LDLR, PCSK9
 Global reach, higher impact
Global reach, higher impact