13.3
Impact Factor
Theranostics 2021; 11(18):8755-8770. doi:10.7150/thno.63446 This issue Cite
Research Paper
Aberrant activation of the CD45-Wnt signaling axis promotes stemness and therapy resistance in colorectal cancer cells
1. School of Life Sciences, Gwangju Institute of Science and Technology, Gwangju 61005, Republic of Korea.
2. Cell Logistics Research Center, Gwangju Institute of Science and Technology, Gwangju 61005, Republic of Korea.
3. Division of Colon and Rectal Surgery, Department of Surgery, Gil Medical Center, Gachon University College of Medicine, Incheon 21999, Republic of Korea.
4. Department of Pathology, College of Medicine, Catholic University of Daegu, College of Medicine, Daegu 38430, Republic of Korea.
5. Department of Pathology, College of Medicine, Chungnam National University, Daejeon 34134, Republic of Korea.
6. Research Institute and Hospital, National Cancer Center, Goyang 10408, Republic of Korea.
7. Drug Information Platform Center, Korea Research Institute of Chemical Technology, Daejeon 34114, Republic of Korea.
Abstract
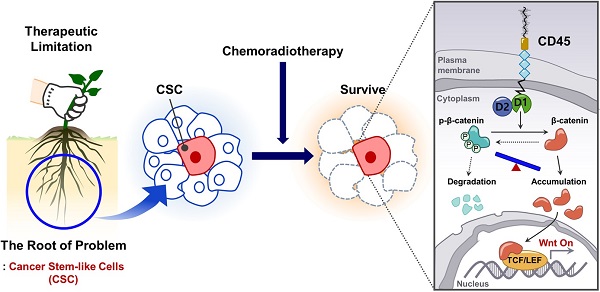
Rationale: Chemoradiation (CRT) is commonly used as an adjuvant or neoadjuvant treatment for colorectal cancer (CRC) patients. However, resistant cells manage to survive and propagate after CRT, increasing the risk of recurrence. Thus, better understanding the mechanism of resistant cancer cells is required to achieve better clinical outcomes.
Methods: Here, we explored gene expression profiling of CRC patient tumors to identify therapy resistance genes and discovered that protein tyrosine phosphatase receptor type C (PTPRC), which encodes CD45, was increased in remnant tumor tissues after CRT and correlated with metastasis. Through multiple validations using patient tumors and CRC cell lines, we found for the first time the increase of CD45 expression in CRC (EpCAM+) epithelial cells surviving after CRT. Thus, we investigated the biological role and downstream events of CD45 were explored in human CRC cells and CRC mouse models.
Results: Increased CD45 expression in cancer cells in pretreated primary tumors accounts for poor regression and recurrence-free survival in CRT-treated patients. High CD45 expression promotes CRC cell survival upon 5-fluorouracil or radiation treatment, while CD45 depletion sensitizes CRC cells to CRT. Intriguingly, CD45 is preferentially expressed in cancer stem-like cells (CSCs), as determined by spheroid culture and the expression of CSC markers, and is required for the distinct functions of CSCs, such as cancer initiation, repopulation, and metastasis. Mechanistically, CD45 phosphatase activity promotes Wnt transcriptional activity by stabilizing the β-catenin protein, which collectively enhances stemness and the therapy-resistant phenotype.
Conclusions: Our results highlight a novel function of CD45 as a mediator of CRT resistance and provide a potential therapy strategy for CRC therapy.
Keywords: Chemoradiation, Colorectal cancer, Therapy resistance, CD45, Cancer stem-like cell, Wnt, β-Catenin
 Global reach, higher impact
Global reach, higher impact