13.3
Impact Factor
Theranostics 2021; 11(17):8185-8196. doi:10.7150/thno.59121 This issue Cite
Research Paper
Designer exosomes for targeted and efficient ferroptosis induction in cancer via chemo-photodynamic therapy
1. Department of Hepatobiliary Surgery, Xijing Hospital, Fourth Military Medical University, Xi'an, 710032, China.
2. State Key Laboratory of Cancer Biology, Biotechnology Center, School of Pharmacy, Fourth Military Medical University, Xi'an, 710032, China.
3. Department of Hematology, Tangdu Hospital, Fourth Military Medical University, Xi'an, 710038, People's Republic of China.
4. Department of Clinical Laboratory, The Second People's Hospital of Hefei, Hefei, China.
5. State Key Laboratory of Cancer Biology, Department of Biochemistry and Molecular Biology, Fourth Military Medical University, Xi'an, 710032, People's Republic of China.
# These authors contributed equally to this article.
Abstract
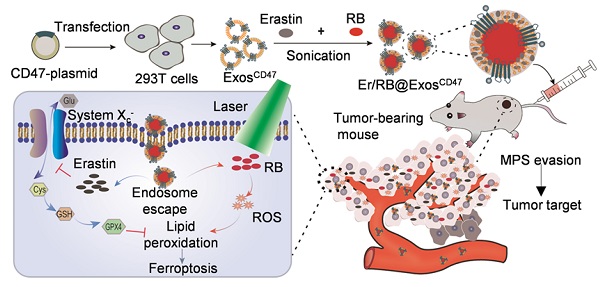
Background: Efficient and specific induction of cell death in liver cancer is urgently needed. In this study, we aimed to design an exosome-based platform to deliver ferroptosis inducer (Erastin, Er) and photosensitizer (Rose Bengal, RB) into tumor tissues with high specificity.
Methods: Exosome donor cells (HEK293T) were transfected with control or CD47-overexpressing plasmid. Exosomes were isolated and loaded with Er and RB via sonication method. Hepa1-6 cell xenograft C57BL/6 model was injected with control and engineered exosomes via tail vein. In vivo distribution of the injected exosomes was analyzed via tracking the fluorescence labeled exosomes. Photodynamic therapy was conducted by 532 nm laser irradiation. The therapeutic effects on hepatocellular carcinoma and toxic side-effects were systemically analyzed.
Results: CD47 was efficiently loaded on the exosomes from the donor cells when CD47 was forced expressed by transfection. CD47 surface functionalization (ExosCD47) made the exosomes effectively escape the phagocytosis of mononuclear phagocyte system (MPS), and thus increased the distribution in tumor tissues. Erastin and RB could be effectively encapsulated into exosomes after sonication, and the drug-loaded exosomes (Er/RB@ExosCD47) strongly induced ferroptosis both in vitro and in vivo in tumor cells after irradiation of 532 nm laser. Moreover, compared with the control exosomes (Er/RB@ExosCtrl), Er/RB@ExosCD47 displayed much lower toxicity in liver.
Conclusion: The engineered exosomes composed of CD47, Erastin, and Rose Bengal, induce obvious ferroptosis in hepatocellular carcinoma (HCC) with minimized toxicity in liver and kidney. The proposed exosomes would provide a promising strategy to treat types of malignant tumors.
Keywords: Hepatocellular carcinoma, exosomes, ferroptosis, photodynamic therapy, synergistic effects
 Global reach, higher impact
Global reach, higher impact