Impact Factor
Theranostics 2019; 9(12):3622-3638. doi:10.7150/thno.32935 This issue Cite
Research Paper
SCARNA10, a nuclear-retained long non-coding RNA, promotes liver fibrosis and serves as a potential biomarker
1. Department of Histology and Embryology, Tianjin Key Laboratory of Cellular and Molecular Immunology, Key Laboratory of Immune Microenvironment and Disease of Ministry of Education, School of Basic Medical Sciences, Tianjin Medical University, Tianjin, China
2. The Third Central Clinical College of Tianjin Medical University, Department of Hepatology and Gastroenterology, Tianjin Third Central Hospital affiliated to Nankai University, Tianjin Key Laboratory of Artificial Cells, Artificial Cell Engineering Technology Research Center of Public Health Ministry, Tianjin, China
3. Department of Immunology, Tianjin Key Laboratory of Cellular and Molecular Immunology, Key Laboratory of Immune Microenvironment and Disease of Ministry of Education, School of Basic Medical Sciences, Tianjin Medical University, Tianjin, China
*These authors contributed equally to this work.
Abstract
Long non-coding RNAs (lncRNAs) are involved in numerous biological functions and pathological processes. However, the clinical significance of lncRNAs and their functions in liver fibrosis remain largely unclear.
Methods: The transcript of lncRNA SCARNA10 in serum and liver samples from patients with advanced hepatic fibrosis, liver tissues from two fibrosis mouse models, and cultured hepatic stellate cells (HSCs) was determined by real-time RT-PCR. The effects of lentivirus-mediated knockdown or over-expression of SCARNA10 in liver fibrosis were examined in vitro and in vivo. Moreover, the effects and mechanisms of down-regulation or over-expression of SCARNA10 on the expression of the genes involved in TGFβ pathway were determined.
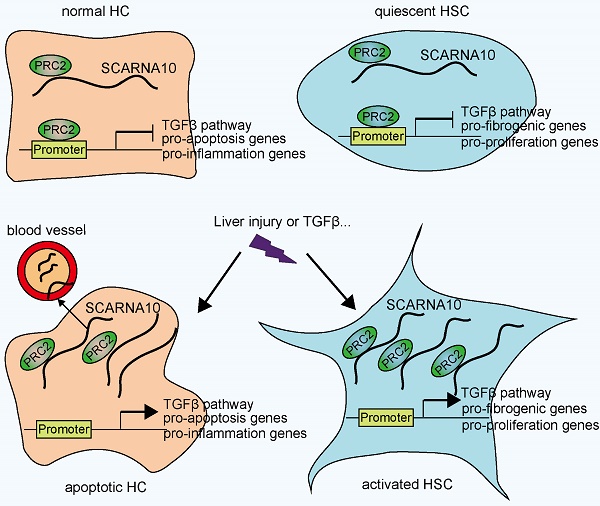
Results: It was found lncRNA ENSMUST00000158992, named as Scarna10, was remarkably up-regulated in mouse fibrotic livers according to the microarray data. We observed that the transcript of SCARNA10 was increased in the serum and liver from patients with advanced hepatic fibrosis. Furthermore, we found that SCARNA10 promoted liver fibrosis both in vitro and in vivo through inducing hepatocytes (HCs) apoptosis and HSCs activation. Mechanistically, RNA immunoprecipitation (RIP) assays demonstrated that SCARNA10 physically associated with polycomb repressive complex 2 (PRC2). Additionally, our results demonstrated that SCARNA10 functioned as a novel positive regulator of TGFβ signaling in hepatic fibrogenesis by inhibiting the binding of PRC2 to the promoters of the genes associated with ECM and TGFβ pathway, thus promoting the transcription of these genes.
Conclusions: Our study identified a crucial role of SCARNA10 in liver fibrosis, providing a proof of this molecule as a potential diagnostic marker and a possible therapeutic target against liver fibrosis.
Keywords: lncRNA, Liver Fibrosis, SCARNA10, TGFβ, PRC2
 Global reach, higher impact
Global reach, higher impact