13.3
Impact Factor
Theranostics 2018; 8(19):5200-5212. doi:10.7150/thno.27806 This issue Cite
Research Paper
Down-regulation of OGT promotes cisplatin resistance by inducing autophagy in ovarian cancer
1. Department of Gynecology and Obstetrics, Xijing Hospital, Fourth Military Medical University, Xi'an, Shaanxi 710032, China;
2. State Key Laboratory of Military Stomatology & National Clinical Research Center for Oral Diseases & Shaanxi International Joint Research Center for Oral Diseases, Center for Tissue Engineering, School of Stomatology, Fourth Military Medical University, Xi'an, Shaanxi 710032, China;
3. Department of Pharmacy, the 456th Hospital of the People's Liberation Army, Jinan Shandong 250031, China;
4. Department of Traditional Chinese Medicine, Xijing Hospital, Fourth Military Medical University, Xi'an, Shaanxi 710032, China;
5. Department of Radiology, Xijing Hospital, Fourth Military Medical University, Xi'an, Shaanxi 710032, China.
#These authors contributed equally to this work.
Abstract
Cisplatin resistance significantly affects the survival rate of patients with ovarian cancer. However, the main mechanism underlying cisplatin resistance in ovarian cancer remains unclear.
Methods: Immunohistochemistry was used to determine the expression of OGT, OGA and O-GlcNAc in chemoresistant and chemosensitive ovarian cancer tissues. Functional analyses (in vitro and in vivo) were performed to confirm the role of OGT in cisplatin resistance. Autophagy-related proteins were tested by western blot. Transmission electron microscopy and mRFP-GFP-LC3 adenovirus reporter were used for autophagy flux analysis. Immunoprecipitation assay was utilized to detect protein-protein interactions.
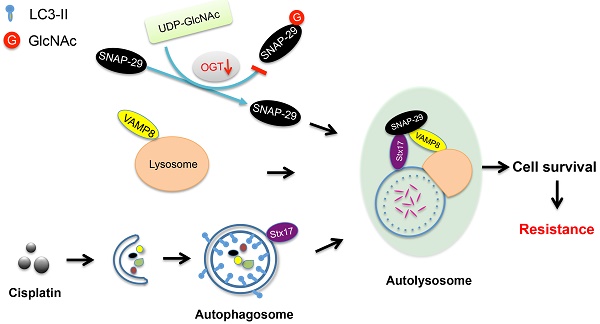
Results: We found that O-GlcNAc and O-GlcNAc transferase (OGT) levels were significantly lower in chemoresistant ovarian cancer tissues than in chemosensitive tissues, whereas O-GlcNAcase (OGA) levels did not differ. The down-regulation of OGT increased cisplatin resistance in ovarian cancer cells but had no effect on the efficacy of paclitaxel. The down-regulation of OGT improved tumor resistance to cisplatin in a mouse xenograft tumor model. OGT knockdown enhanced cisplatin-induced autophagy, which reduced apoptotic cell death induced by cisplatin, and promoted autolysosome formation. A reduction in O-GlcNAcylated SNAP-29 levels caused by the down-regulation of OGT promoted the formation of the SNARE complex and autophagic flux.
Conclusion: Our findings suggest that down-regulation of OGT enhances cisplatin-induced autophagy via SNAP-29, resulting in cisplatin-resistant ovarian cancer. OGT may represent a novel target for overcoming cisplatin resistance in ovarian cancer.
Keywords: autophagy, chemoresistance, OGT, ovarian cancer, SNARE.
 Global reach, higher impact
Global reach, higher impact