13.3
Impact Factor
Theranostics 2023; 13(15):5546-5560. doi:10.7150/thno.87340 This issue Cite
Research Paper
Engineered oncolytic bacteria HCS1 exerts high immune stimulation and safety profiles for cancer therapy
1. School of Biomedical Science, Hunan University, Changsha 410082, China.
2. College of Biology, Hunan University, Changsha 410082, China.
3. Department of Neurosurgery, Guangzhou First People′s Hospital, Guangzhou 510180, China.
4. Department of Nuclear Medicine, the Second Affiliated Hospital of Guangzhou Medical University, Guangzhou, 510260, China.
5. School of Food Science and Bioengineering, Changsha University of Science & Technology, Changsha, Hunan 410114, China.
Abstract
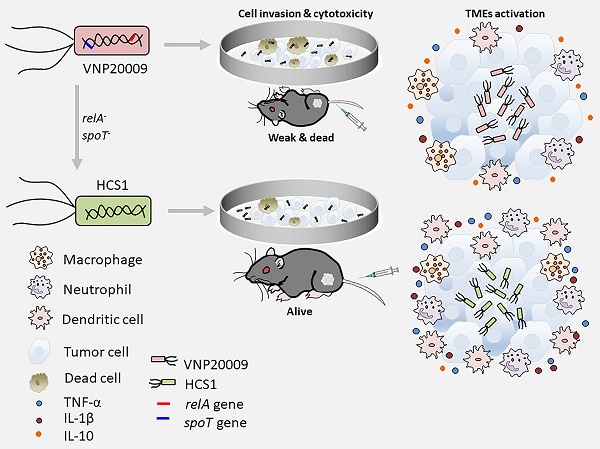
Background and rationale: Attenuated Salmonella typhimurium VNP20009 has been used to treat tumor-bearing mice and entered phase I clinical trials. However, its mild anticancer effect in clinical trials may be related to insufficient bacterial colonization and notable adverse effects with increasing dosages. Guanosine 5′-diphosphate-3′-diphosphate (ppGpp) synthesis-deficient Salmonella is an attenuated strain with good biosafety and anticancer efficacy that has been widely investigated in various solid cancers in preclinical studies. Integration of the advantages of these two strains may provide a new solution for oncolytic bacterial therapy.
Methods: We incorporated the features of ΔppGpp into VNP20009 and obtained the HCS1 strain by deleting relA and spoT, and then assessed its cytotoxicity in vitro and antitumor activities in vivo.
Results: In vitro experiments revealed that the invasiveness and cytotoxicity of HCS1 to cancer cells were significantly lower than those of the VNP20009. Additionally, tumor-bearing mice showed robust cancer suppression when treated with different doses of HCS1 intravenously, and the survival time and cured mice were dramatically increased. Furthermore, HCS1 can increase the levels of pro-inflammatory cytokines in tumor tissues and relieve the immunosuppression in the tumor microenvironments. It can also recruit abundant immune cells into tumor tissues, thereby increasing immune activation responses.
Conclusion: The newly engineered Salmonella HCS1 strain manifests high prospects for cancer therapeutics and is a promising option for future clinical cancer immunotherapy.
Keywords: bacterial-mediated cancer therapy, Salmonella typhimurium, VNP20009, systemic safety, clinical translation
 Global reach, higher impact
Global reach, higher impact