13.3
Impact Factor
Theranostics 2023; 13(7):2072-2087. doi:10.7150/thno.81198 This issue Cite
Research Paper
Transcription factor TOX maintains the expression of Mst1 in controlling the early mouse NK cell development
1. The Fifth Affiliated Hospital (Heyuan Shenhe People's Hospital), Jinan University, Heyuan 517000, China
2. The Biomedical Translational Research Institute, Guangzhou Key Laboratory for Germ-free animals and Microbiota Application, Key Laboratory of Ministry of Education for Viral Pathogenesis & Infection Prevention and Control, School of Medicine, Jinan University, Guangzhou, 510632, China
3. Guangdong Provincial Key Laboratory of Tumor Interventional Diagnosis and Treatment, Zhuhai Institute of Translational Medicine, Zhuhai People's Hospital Affiliated with Jinan University, Jinan University, Zhuhai, 519000, China
4. Department of Orthopedics, The First Affiliated Hospital, Jinan University, Guangzhou, 510630, China
5. The First Affiliated Hospital of Anhui Medical University and Institute for Clinical Immunology, Anhui Medical University, Anhui, 230032, China
6. School of Medicine and Institute for Immunology, Tsinghua University, Beijing, 100084, China
*These authors contributed equally to this work
Abstract
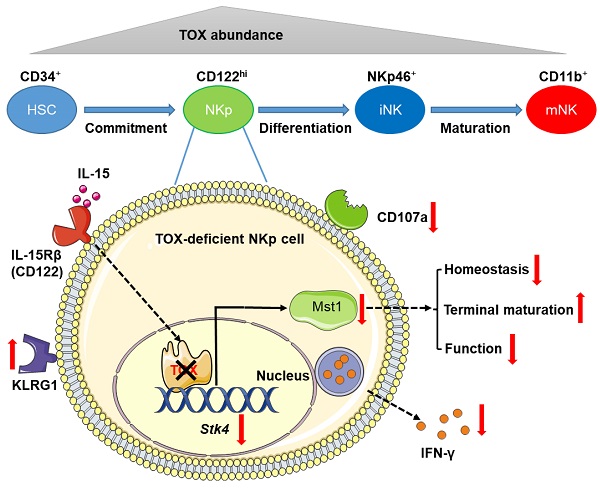
Rationale: TOX is a DNA-binding factor required for the development of multiple immune cells and the formation of lymph nodes. However, the temporal regulation mode of TOX on NK cell development and function needs to be further explored.
Methods: To investigate the role of TOX in NK cells at distinct developmental phases, we deleted TOX at the hematopoietic stem cell stage (Vav-Cre), NK cell precursor (CD122-Cre) stage and late NK cell developmental stage (Ncr1-Cre), respectively. Flow cytometry was used to detect the development and functional changes of NK cell when deletion of TOX. RNA-seq was used to assess the differences in transcriptional expression profile of WT and TOX-deficient NK cells. Published Chip-seq data was exploited to search for the proteins directly interact with TOX in NK cells.
Results: The deficiency of TOX at the hematopoietic stem cell stage severely retarded NK cell development. To a less extent, TOX also played an essential role in the physiological process of NKp cells differentiation into mature NK cells. Furthermore, the deletion of TOX at NKp stage severely impaired the immune surveillance function of NK cells, accompanied by down-regulation of IFN-γ and CD107a expression. However, TOX is dispensable for mature NK cell development and function. Mechanistically, by combining RNA-seq data with published TOX ChIP-seq data, we found that the inactivation of TOX at NKp stage directly repressed the expression of Mst1, an important intermediate kinase in Hippo signaling pathway. Mst1 deficient at NKp stage gained the similar phenotype with Toxfl/flCD122Cre mice.
Conclusion: In our study, we conclude that TOX coordinates the early mouse NK cell development at NKp stage by maintaining the expression of Mst1. Moreover, we clarify the different dependence of the transcription factor TOX in NK cells biology.
Keywords: TOX, NK cell, Mst1, cell development, immune surveillance
 Global reach, higher impact
Global reach, higher impact