Impact Factor
Theranostics 2022; 12(17):7420-7430. doi:10.7150/thno.77761 This issue Cite
Research Paper
Identification of a miRNA-based non-invasive predictive biomarker of response to target therapy in BRAF-mutant melanoma
1. Department of Melanoma, Oncologic Immunotherapy and Innovative Therapies, Istituto Nazionale Tumori-IRCCS-Fondazione G. Pascale, Napoli, Italy.
2. SAFU Laboratory, Department of Research, Advanced Diagnostics and Technological Innovation, Translational Research Area, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
3. UOSD Clinical Trial Center e Biostatistica e Bioinformatica, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
4. San Gallicano Dermatological Institute IRCCS UOSD Clinical Trial Center e Biostatistica e Bioinformatica, Scientific Direction Via Elio Chianesi 53, 00144, Rome, Italy
5. Department. Preclinical Models and New Therapeutic Agents Unit, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
6. Oncogenomic and Epigenetic Unit, Department of Diagnostic Research and Technological Innovation, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
7. Biological Tissue and Liquid Bank, Scientific Direction, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
8. Clinical Pathology, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
9. Department of Molecular and Clinical Medicine, University of Rome “Sapienza”, 00161 Rome, Italy.
10. Department of Clinical and Molecular Medicine, Sapienza University of Rome, Rome, Italy.
11. Division of Medical Oncology1, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
12. Department of Experimental and Clinical Medicine, University “Magna Graecia” of Catanzaro, Catanzaro, Italy.
13. Scientific Direction, IRCCS Regina Elena National Cancer Institute, Rome, Italy.
† co-authors
Abstract
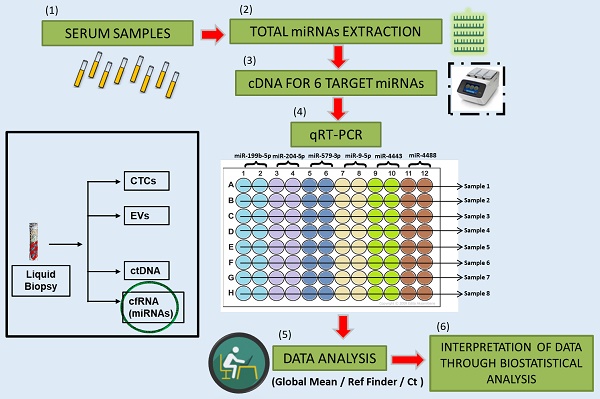
Rationale: Metastatic melanoma is the most aggressive and dangerous form of skin cancer. The introduction of immunotherapy with Immune checkpoint Inhibitors (ICI) and of targeted therapy with BRAF and MEK inhibitors for BRAF mutated melanoma, has greatly improved the clinical outcome of these patients. Nevertheless, response to therapy remains highly variable and the development of drug resistance continues to be a daunting challenge. Within this context there is a need to develop diagnostic tools capable of predicting response or resistance to therapy in order to select the best therapeutic approach. Over the years, accumulating evidence brought to light the role of microRNAs (miRNAs) as disease biomarkers.
Methods: In particular, the detection of miRNAs in whole blood or specific blood components such as serum or plasma, allows these molecules to be good candidates for diagnosis, prognosis and for monitoring response to anticancer therapy. In this paper, we evaluated circulating basal levels of 6 previously identified miRNAs in serum samples of 70 BRAF-mutant melanoma patients before starting targeted therapy.
Results: Results show that the circulating levels of the oncosuppressor miR-579-3p and of the oncomiR miR-4488 are able to predict progression free survival (PFS) but not overall survival (OS). Most importantly, we observed that the best predictor of disease outcome is represented by the ratio of circulating miR-4488 vs. miR-579-3p (miRatio). Finally, the combination of the Lactate dehydrogenase (LDH) blood levels with the two circulating miRNAs alone or together did not produce any improvement in predicting PFS indicating that miR-579-3p and miR-4488 are independent predictors of PFS as compared to LDH.
Conclusions: All together these data underscored the relevance of circulating miRNAs as suitable tools to predict therapy response in melanoma and maybe further developed as companion diagnostics in the clinic.
Keywords: microRNA, liquid biopsy, biomarkers, metastatic melanoma, target therapy.
 Global reach, higher impact
Global reach, higher impact