13.3
Impact Factor
Theranostics 2022; 12(13):5836-5855. doi:10.7150/thno.73400 This issue Cite
Review
Extracellular Vesicles and Circulating Tumour Cells - complementary liquid biopsies or standalone concepts?
1. Department of Pathophysiology, Nicolaus Copernicus University in Toruń, Ludwik Rydygier Collegium Medicum in Bydgoszcz, 85-067 Bydgoszcz, Poland.
2. Department of Internal Medicine I, University Hospital Bonn of the Rheinische Friedrich-Wilhelms-University, 53127 Bonn, Germany.
3. Institute of Molecular Medicine & Experimental Immunology, University Hospital Bonn of the Rheinische Friedrich-Wilhelms-University, 53127 Bonn, Germany.
4. Octavian Fodor Institute for Gastroenterology and Hepatology, Iuliu Haţieganu, University of Medicine and Pharmacy, 400162 Cluj-Napoca, Romania.
5. Department of General, Visceral and Vascular Surgery, German Armed Forces Hospital Hamburg, 22049 Hamburg, Germany.
6. Department of Integrated Oncology, Center for Integrated Oncology (CIO), University Hospital Bonn of the Rheinische Friedrich-Wilhelms-University, 53127 Bonn, Germany.
* Shared first author
Abstract
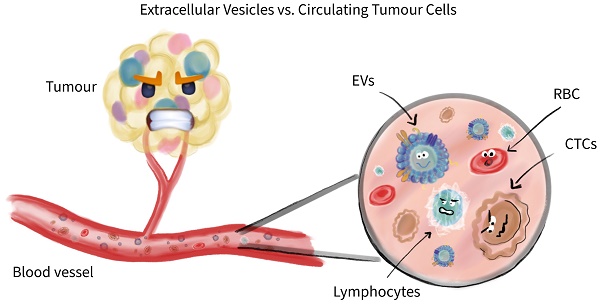
Liquid biopsies do promise a lot, but are they keeping it? In the past decade, additional novel biomarkers qualified to be called like that, of which, some took necessary hurdles resulting in FDA approval and clinical use. Some others are since a while around, well known and were once regarded to be a game changer in cancer diagnosis or cancer screening. But, during their clinical use limitations were observed from statistical significance and questions raised regarding their robustness, that eventually led to be dropped from associated clinical guidelines for certain applications including cancer diagnosis. The purpose of this review isn't to give a broad overview of all current liquid biopsy as biomarkers, weight them and promise a brighter future in cancer prevention, but rather to take a deeper look on two of those who do qualify to be called liquid biopsies now or then. These two are probably of greatest interest conceptually and methodically, and likely have the highest chances to be in clinical use soon, with a portfolio extension over their original conceptual usage. We aim to dig deeper beyond cancer diagnosis or cancer screening. Actually, we aim to review in depth extracellular vesicles (EVs) and compare with circulating tumour cells (CTCs). The latter methodology is partially FDA approved and in clinical use. We will lay out similarities as taking advantage of surface antigens on EVs and CTCs in case of characterization and quantification. But drawing readers' attention to downstream application based on capture/isolation methodology and simply on their overall nature, here apparently being living material eventually recoverable as CTCs are vs. dead material with transient effects on recipient cell as in case of EVs. All this we try to bring in perspective, compare and conclude towards which future direction we are aiming for, or should aim for. Do we announce a winner between CTCs vs EVs? No, but we provide good reasons to intensify research on them.
Keywords: CTC, extracellular vesicles, exosomes, microvesicles, ectosomes, biomarker, liquid biopsy, personalized medicine
 Global reach, higher impact
Global reach, higher impact