Impact Factor
Theranostics 2022; 12(11):4935-4948. doi:10.7150/thno.73223 This issue Cite
Review
Histone acetyltransferases CBP/p300 in tumorigenesis and CBP/p300 inhibitors as promising novel anticancer agents
1. Department of Oncology, 3201 Hospital of Xi'an Jiaotong University Health Science Center, Hanzhong, Shaanxi 723000, China.
2. School of Medicine and Public Health, Priority Research Centre for Cancer Research, University of Newcastle, Callaghan, Newcastle, NSW 2308, Australia.
3. Translational Research Institute, Henan Provincial People's Hospital, Academy of Medical Sciences, Zhengzhou University, Zhengzhou, China.
4. Department of Pharmacology, School of Basic Medical Sciences, Zhengzhou University, Zhengzhou, China.
5. Children's Cancer Institute Australia, Randwick, Sydney, NSW 2031, Australia.
6. School of Women's and Children's Health, University of New South Wales, Sydney, New South Wales, Australia.
Abstract
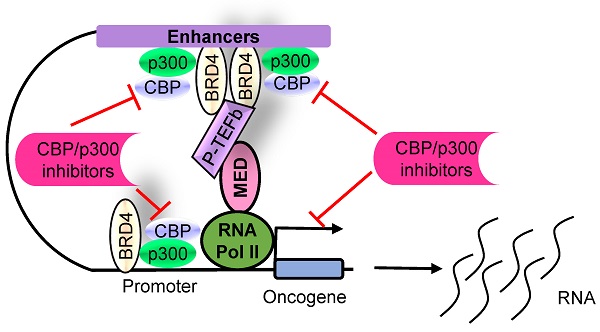
The histone acetyltransferases CBP and p300, often referred to as CBP/p300 due to their sequence homology and functional overlap and co-operation, are emerging as critical drivers of oncogenesis in the past several years. CBP/p300 induces histone H3 lysine 27 acetylation (H3K27ac) at target gene promoters, enhancers and super-enhancers, thereby activating gene transcription. While earlier studies indicate that CBP/p300 deletion/loss can promote tumorigenesis, CBP/p300 have more recently been shown to be over-expressed in cancer cells and drug-resistant cancer cells, activate oncogene transcription and induce cancer cell proliferation, survival, tumorigenesis, metastasis, immune evasion and drug-resistance. Small molecule CBP/p300 histone acetyltransferase inhibitors, bromodomain inhibitors, CBP/p300 and BET bromodomain dual inhibitors and p300 protein degraders have recently been discovered. The CBP/p300 inhibitors and degraders reduce H3K27ac, down-regulate oncogene transcription, induce cancer cell growth inhibition and cell death, activate immune response, overcome drug resistance and suppress tumor progression in vivo. In addition, CBP/p300 inhibitors enhance the anticancer efficacy of chemotherapy, radiotherapy and epigenetic anticancer agents, including BET bromodomain inhibitors; and the combination therapies exert substantial anticancer effects in mouse models of human cancers including drug-resistant cancers. Currently, two CBP/p300 inhibitors are under clinical evaluation in patients with advanced and drug-resistant solid tumors or hematological malignancies. In summary, CBP/p300 have recently been identified as critical tumorigenic drivers, and CBP/p300 inhibitors and protein degraders are emerging as promising novel anticancer agents for clinical translation.
Keywords: CBP/p300, gene transcription, tumorigenesis, small molecule inhibitors, cancer therapy
 Global reach, higher impact
Global reach, higher impact