Impact Factor
Theranostics 2022; 12(7):3438-3455. doi:10.7150/thno.61433 This issue Cite
Research Paper
Cathelicidin-related antimicrobial peptide protects against enteric pathogen-accelerated type 1 diabetes in mice
1. Wuxi School of Medicine and School of Food Science and Technology, Jiangnan University, Wuxi 214122, Jiangsu, P. R. China.
2. State Key Laboratory of Food Science and Technology, Jiangnan University, Wuxi 214122, Jiangsu, P. R. China.
3. College of Food and Pharmaceutical Sciences, Ningbo University, Ningbo 315800, Zhejiang, P. R. China.
4. CAS Key Laboratory of Tissue Microenvironment and Tumor, Shanghai Institute of Nutrition and Health, Shanghai Institutes for Biological Sciences, University of Chinese Academy of Sciences, Chinese Academy of Sciences, Shanghai 200031, P. R. China.
*These authors contributed equally to this work.
Abstract
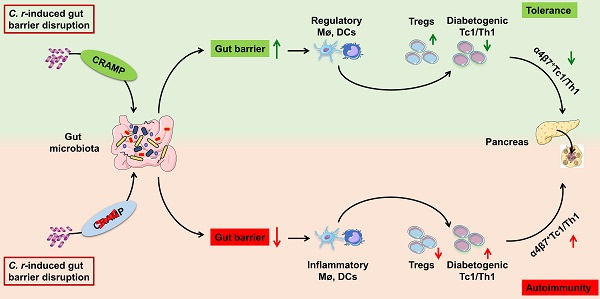
Rationale: Gut barrier disruption caused by enteric pathogen infection results in activated diabetogenic T cells and accelerated type 1 diabetes (T1D). Cathelicidin-related antimicrobial peptide (CRAMP) maintains intestinal barrier integrity, regulates the microbiome, and exerts positive immune-modulatory effects on pancreatic diseases.
Methods: The model enteric pathogen Citrobacter rodentium (C. rodentium) was adopted to represent clinical colonic infection with gut barrier disruption. The protective role and gut-pancreas pathophysiological mechanism of CRAMP in enteric pathogen-accelerated T1D were investigated in spontaneous non-obese diabetic (NOD) mice and streptozotocin-induced diabetic mice.
Results: Colonic CRAMP production was defective in C. rodentium infection-accelerated T1D. C. rodentium infection triggered the recruitment of interferon-gamma (IFN-γ)+ T cells and accelerated T1D. In the C. rodentium-accelerated T1D mice, CRAMP deficiency further aggravated gut barrier disruption, gut dysbiosis, and diabetic phenotype, which could be reversed by CRAMP treatment. The protective effect of CRAMP may be due to CRAMP inhibiting C. rodentium-aggravated gut immune dysregulation, gut dysbiosis, and migration of gut-primed IFN-γ+ T cells to the pancreas, thus contributing to gut barrier protection and pancreatic-intestinal immune homeostasis.
Conclusion: CRAMP plays a pivotal role in pancreatic-gut crosstalk during C. rodentium-accelerated T1D by gut barrier-protective, immune- and microbial-modulatory mechanisms. Cathelicidin supplementation to restore a healthy gut barrier may represent a novel therapeutic strategy for T1D.
Keywords: antimicrobial peptides, gut barrier, type 1 diabetes, gut-pancreas crosstalk, intestinal IFNγ+ T cell migration
 Global reach, higher impact
Global reach, higher impact