13.3
Impact Factor
Theranostics 2022; 12(7):3251-3272. doi:10.7150/thno.70623 This issue Cite
Research Paper
Mitochondria-targeted supramolecular coordination container encapsulated with exogenous itaconate for synergistic therapy of joint inflammation
1. Department of Oral Surgery, Shanghai Key Laboratory of Stomatology & Shanghai Research Institute of Stomatology, National Clinical Research Center for Oral Diseases, Shanghai Ninth People's Hospital, College of Stomatology, Shanghai Jiao Tong University School of Medicine, Shanghai, 200011, China
2. State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou, Fujian, 350002, China
3. Department of Orthopedics, Shanghai Key Laboratory of Orthopedic Implant, Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, 200011, China
4. Department of Chemistry & Center for Fluorinated Functional Materials, University of South Dakota, Vermillion, South Dakota, 57069-2390, United States
*These authors contributed equally.
Abstract
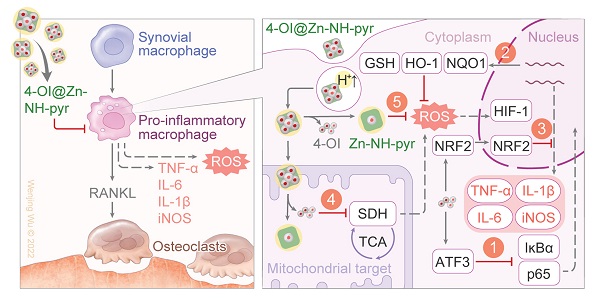
Rationale: Inflammatory macrophages and osteoclasts (OCs) play critical roles in joint inflammation, which feature the excessive production of reactive oxygen species (ROS), resulting in synovial inflammation and bone erosion. Scavenging ROS, especially by modulating mitochondrial metabolic activity, could be a desirable strategy for the management of inflammatory joints. This study aimed to develop a mitochondria-targeted supramolecular drug delivery system with exogenous and endogenous ROS-scavenging activities for the treatment of joint inflammation.
Methods: In this study, we utilized a zinc-based metal-organic supercontainer (MOSC) as a proton sponge and electron reservoir with outstanding proton binding capacity, extracellular ROS-scavenging ability, and biocompatibility to establish an efficient supramolecular nanocarrier for endo/lysosomal escape and mitochondrial targeting. 4-Octyl itaconate (4-OI), an itaconate derivative, served as the loaded guest for the construction of a synergistic therapeutic system for inflammatory macrophages and OCs.
Results: After the effective encapsulation of 4-OI, 4-OI@Zn-NH-pyr not only exhibited potent ROS-scavenging capacity, but also reduced ROS production by mediating mitochondrial respiration in inflammatory macrophages. Regarding its anti-inflammatory efficacy, 4-OI@Zn-NH-pyr ameliorated the inflammatory reaction by activating nuclear factor erythroid 2-related factor 2 (Nrf2), thus increasing the production of antioxidants, apart from the inhibition of NF-κB pathways. Additionally, receptor activator of nuclear factor-κB ligand (RANKL)-induced osteoclast differentiation and function was remarkably suppressed by 4-OI@Zn-NH-pyr. Consistent with in vitro observations, 4-OI@Zn-NH-pyr efficiently inhibited synovial inflammation and subchondral bone destruction in an acute arthritis model.
Conclusion: By using MOSCs that are highly reactive to ROS as drug-loaded matrices for the first time, this study provides an avenue for the management of severe joint inflammation by designing synergistic supramolecular drug-delivery systems with subcellular targeting and ROS-scavenging capacity.
Keywords: Coordination containers, Supermolecule, ROS scavenging, Joint inflammation, Exogenous itaconate
 Global reach, higher impact
Global reach, higher impact