13.3
Impact Factor
Theranostics 2022; 12(2):875-890. doi:10.7150/thno.66420 This issue Cite
Research Paper
5-aza-2′-deoxycytidine potentiates anti-tumor immunity in colorectal peritoneal metastasis by modulating ABC A9-mediated cholesterol accumulation in macrophages
1. Department of Biochemistry and Molecular Biology, Third Military Medical University (Army Medical University), Chongqing 400038, China.
2. Chongqing Weisiteng Biotech Translational Research Institute, Chongqing 400039, China.
3. Department of General Surgery, Southwest Hospital, Third Military Medical University (Army Medical University), Chongqing 400038, China.
Abstract
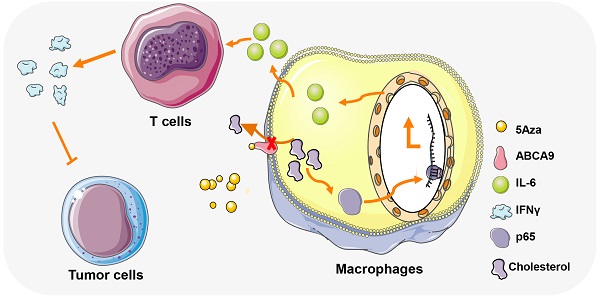
Background: 5-aza-2'-deoxycytidine (5Aza), a DNA methyltransferase (DNMT) inhibitor, could activate tumor adaptive immunity to inhibit tumor progression. However, the molecular mechanisms by which 5Aza regulates tumor immune microenvironment are still not fully understood.
Methods: The role of 5Aza in immune microenvironment of peritoneal carcinomatosis (PC) of colorectal cancer (CRC) was investigated. The effects of 5Aza on macrophage activation were studied by flow cytometry, real-time PCR, Western blotting assays, and Drug Affinity Responsive Target Stability (DARTS). The effects of 5Aza on tumor immunity were validated in stromal macrophages and T cells from CRC patients.
Results: 5Aza could stimulate the activation of macrophages toward an M1-like phenotype and subsequent activation of T cells in premetastatic fat tissues, and ultimately suppress CRC-PC in immune-competent mouse models. Mechanistically, 5Aza stimulated primary mouse macrophages toward to a M1-like phenotype characterized by the increase of p65 phosphorylation and IL-6 expression. Furthermore, we screened and identified ATP-binding cassette transporter A9 (ABC A9) as a binding target of 5Aza. 5Aza induced cholesterol accumulation, p65 phosphorylation and IL-6 expression in an ABC A9-dependent manner. Pharmacological inhibition of NF-κB, or genetic depletion of IL-6 abolished the antitumor effect of 5Aza in mice. In addition, the antitumor effect of 5Aza was synergistically potentiated by conventional chemotherapeutic drugs 5-Fu or OXP. Finally, we validated the reprogramming role of 5Aza in antitumor immunity in stromal macrophages and T cells from CRC patients.
Conclusions: Taken together, our findings showed for the first time that 5Aza suppressed CRC-PC by regulating macrophage-dependent T cell activation in premetastatic microenvironment, meanwhile uncovered a DNA methylation-independent mechanism of 5Aza in regulating ABC A9-associated cholesterol metabolism and macrophage activation.
Keywords: 5Aza, CRC-PC, macrophages, ABC A9, IL-6
 Global reach, higher impact
Global reach, higher impact