Impact Factor
Theranostics 2022; 12(1):379-395. doi:10.7150/thno.67034 This issue Cite
Research Paper
Smad3 deficiency improves islet-based therapy for diabetes and diabetic kidney injury by promoting β cell proliferation via the E2F3-dependent mechanism
1. Research Center for Integrated Medicine and Department of Nephrology, Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University, Luzhou, Sichuan, 646000, China.
2. Department of Medicine and Therapeutics, and Li Ka Shing Institute of Health Sciences, the Chinese University of Hong Kong, Hong Kong, 999077, China.
3. Guangdong-Hong Kong Joint Laboratory on Immunological and Genetic Kidney Diseases, Guangdong Provincial People's Hospital, Guangdong Academy of Medical Sciences, Guangzhou, Guangdong, 510080, China.
4. State Key Laboratory of Bioelectronics, Jiangsu Key Laboratory for Biomaterials and Devices, School of Biological Sciences & Medical Engineering, Southeast University, Nanjing, China.
5. Key Laboratory of Prevention and Management of Chronic Kidney Disease of Zhanjiang City, Institute of Nephrology, Affiliated Hospital of Guangdong Medical University, Zhanjiang, Guangdong, 524001, China.
6. National Traditional Chinese Medicine Clinical Research Base, Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University, Luzhou, Sichuan, 646000, China.
7. CUHK-Guangdong Provincial People's Hospital Joint Research Laboratory on Immunological and Genetic Kidney Diseases, the Chinese University of Hong Kong, Hong Kong, 999077, China.
Abstract
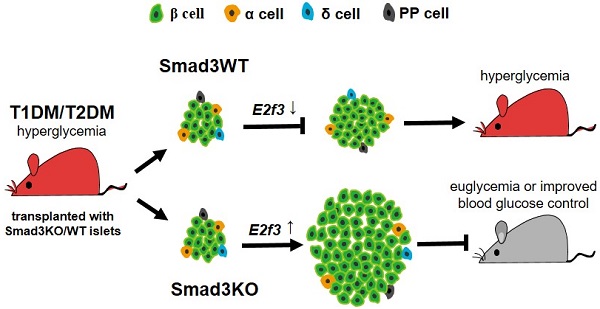
Rationale: Poor β cell proliferation is one of the detrimental factors hindering islet cell replacement therapy for patients with diabetes. Smad3 is an important transcriptional factor of TGF-β signaling and has been shown to promote diabetes by inhibiting β cell proliferation. Therefore, we hypothesize that Smad3-deficient islets may be a novel cell replacement therapy for diabetes.
Methods: We examined this hypothesis in streptozocin-induced type-1 diabetic mice and type-2 diabetic db/db mice by transplanting Smad3 knockout (KO) and wild type (WT) islets under the renal capsule, respectively. The effects of Smad3KO versus WT islet replacement therapy on diabetes and diabetic kidney injury were examined. In addition, RNA-seq was applied to identify the downstream target gene underlying Smad3-regulated β cell proliferation in Smad3KO-db/db versus Smad3WT-db/db mouse islets.
Results: Compared to Smad3WT islet therapy, treatment with Smad3KO islets produced a much better therapeutic effect on both type-1 and type-2 diabetes by significantly lowering serum levels of blood glucose and HbA1c and protected against diabetic kidney injuries by preventing an increase in serum creatinine and the development of proteinuria, mesangial matrix expansion, and fibrosis. These were associated with a significant increase in grafted β cell proliferation and blood insulin levels, resulting in improved glucose intolerance. Mechanistically, RNA-seq revealed that compared with Smad3WT-db/db mouse islets, deletion of Smad3 from db/db mouse islets markedly upregulated E2F3, a pivotal regulator of cell cycle G1/S entry. Further studies found that Smad3 could bind to the promoter of E2F3, and thus inhibit β cell proliferation via an E2F3-dependent mechanism as silencing E2F3 abrogated the proliferative effect on Smad3KO β cells.
Conclusion: Smad3-deficient islet replacement therapy can significantly improve both type-1 and type-2 diabetes and protect against diabetic kidney injury, which is mediated by a novel mechanism of E2F3-dependent β cell proliferation.
Keywords: Smad3, islet transplantation, diabetes, E2F3, cell cycle
 Global reach, higher impact
Global reach, higher impact