13.3
Impact Factor
Theranostics 2022; 12(1):59-75. doi:10.7150/thno.64862 This issue Cite
Research Paper
A hydrogen peroxide economizer for on-demand oxygen production-assisted robust sonodynamic immunotherapy
1. Department of Ultrasound, Chongqing Key Laboratory of Ultrasound Molecular Imaging, the Second Affiliated Hospital of Chongqing Medical University, Chongqing 400010, P. R. China.
2. Department of Medical Ultrasonics, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, 510080, P. R. China.
3. Department of Ultrasound China-Japan Union Hospital of Jilin University, Jilin 130033, P. R. China.
4. Department of Pathology, Chongqing Medical University, Chongqing, 400016, P. R. China.
5. Department of Ultrasound, the First Affiliated Hospital of Chongqing Medical University, Chongqing 400010, China.
6. Department of Ultrasound, the Third People's Hospital of Chengdu City, the Affiliated Hospital of Southwest Jiaotong University.
*Qinqin Jiang and Bin Qiao are co-first authors who contributed equally to this work.
Abstract
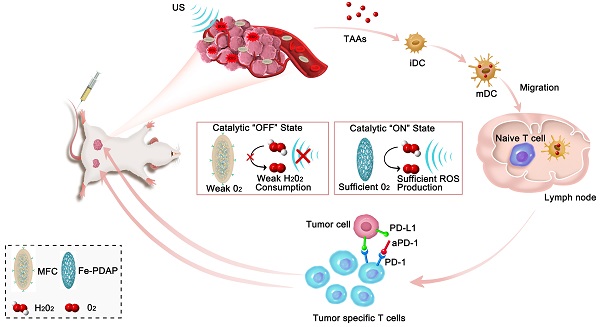
The outcome of sonodynamic immunotherapy is significantly limited by tumor hypoxia. To overcome this obstacle, one common solution is to catalyze the conversion of endogenous H2O2 into O2. However, the effectiveness of this strategy is limited by the insufficient concentration of H2O2 in the tumor microenvironment (TME). Herein, we developed a H2O2 economizer for on-demand O2 supply and sonosensitizer-mediated reactive oxygen species production during ultrasound activation, thereby alleviating hypoxia-associated limitations and augmenting the efficacy of sonodynamic immunotherapy.
Methods: The H2O2 economizer is constructed by electrostatic adsorption and π-π interactions between the Fe-doped polydiaminopyridine (Fe-PDAP) nanozyme and chlorin e6. By employing a biomimetic engineering strategy with cancer cell membranes, we addressed the premature leakage issue and increased tumor-site accumulation of nanoparticles (membrane-coated Fe-PDAP/Ce6, MFC).
Results: The prepared MFC could significantly attenuate the catalytic activity of Fe-PDAP by reducing their contact with H2O2. Ultrasound irradiation promoted MFC dissociation and the exposure of Fe-PDAP for a more robust O2 supply. Moreover, the combination of MFC-enhanced sonodynamic therapy with anti-programmed cell death protein-1 antibody (aPD-1) immune checkpoint blockade induced a strong antitumor response against both primary tumors and distant tumors.
Conclusion: This as-prepared H2O2 economizer significantly alleviates tumor hypoxia via reducing H2O2 expenditure and that on-demand oxygen-elevated sonodynamic immunotherapy can effectively combat tumors.
Keywords: sonodynamic therapy, immunotherapy, nanozyme, cancer cell membrane, focused ultrasound
 Global reach, higher impact
Global reach, higher impact