13.3
Impact Factor
Theranostics 2021; 11(19):9652-9666. doi:10.7150/thno.64354 This issue Cite
Research Paper
Polyamino acid calcified nanohybrids induce immunogenic cell death for augmented chemotherapy and chemo-photodynamic synergistic therapy
1. Key Laboratory of Luminescence Analysis and Molecular Sensing (Southwest University), Ministry of Education, School of Materials and Energy and Chongqing Engineering Research Center for Micro-Nano Biomedical Materials and Devices, Southwest University, Chongqing 400715, P. R. China.
2. Pediatric Research Institute, Department of Hematology and Oncology, Shenzhen Children's Hospital, Shenzhen, Guangdong 518038, P. R. China.
3. Key Laboratory of Laser Technology and Optoelectronic Functional Materials of Hainan Province, College of Chemistry and Chemical Engineering, Hainan Normal University, Haikou 571158, P. R. China.
Abstract
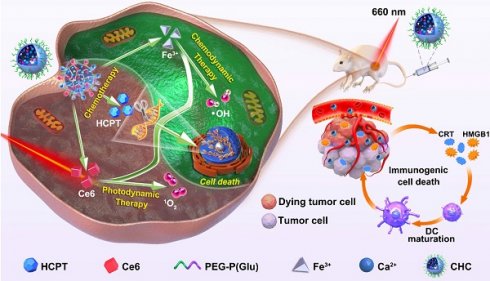
Background: Monotherapy for cancer treatment is limited by unstable efficacy and uncontrollable toxic side effects, while the multifunctional nanoplatform with complex preparation process cannot avoid the potential toxicity of each functional component.
Methods: We exploited tumor-specific activated polyamino acid calcified nanoparticles (CHC NPs) as new-type oxidative stress amplification of anticancer drugs via building a safe and biodegradable multifunctional nanoplatform. Giving priority to chemotherapy, and synergizing chemodynamic therapy (CDT) with photodynamic therapy (PDT), this strategy was to achieve enhanced chemotherapy, simultaneously inducing immunogenic cell death and inhibiting tumor cell invasion.
Results: Based on amorphous calcium carbonate, pH-responsive nanocarrier was prepared with classical chemotherapeutic drug 10-hydroxycamplothecin (HCPT) and photosensitizer Chlorin e6 (Ce6) to realize multifunctional nanotheranostics.
Conclusion: Inventive calcified nanohybrids, where topoisomerase inhibited by HCPT to prevent DNA synthesis, the generation of •OH induced via Fenton reaction, along with a large amount of 1O2 produced by Ce6, might be a promising strategy for anti-tumor combination therapy in clinical translation.
Keywords: Calcified nanohybrids, multimodal nanotheranostics, chemotherapy, chemo-photodynamic therapy, immunogenic cell death
 Global reach, higher impact
Global reach, higher impact