13.3
Impact Factor
Theranostics 2021; 11(12):6006-6018. doi:10.7150/thno.59841 This issue Cite
Research Paper
The kynurenine derivative 3-HAA sensitizes hepatocellular carcinoma to sorafenib by upregulating phosphatases
1. Basic Medical Institute; Hongqiao International Institute of Medicine, Tongren Hospital; Key Laboratory of Cell Differentiation and Apoptosis of Chinese Ministry of Education, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China.
2. Department of Nuclear Medicine & Department of Oncology, Rui Jin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China.
3. Shanghai Ninth People's Hospital, College of Stomatology, Shanghai Jiao Tong University School of Medicine, Shanghai, 200011, China.
4. Translational Medical Center for Stem Cell Therapy, East Hospital, Tongji University School of Medicine, 150 Jimo Road, Shanghai 200120, China.
5. International Co-operation Laboratory on Signal Transduction, Eastern Hepatobiliary Surgery Institute, Marine Military Medical University, Shanghai 200438, China.
6. Department of Liver Surgery, Liver Cancer Institute, Zhongshan Hospital, Fudan University, Shanghai 200032, China.
7. Institute of Cancer Stem Cell, Dalian Medical University, Dalian, Liaoning 116044, China.
8. Department of Gastroenterology, Shanghai Pudong Hospital, Fudan University Pudong Medical Center, Shanghai 201399, China.
*Authors contribute to this paper equally.
Abstract
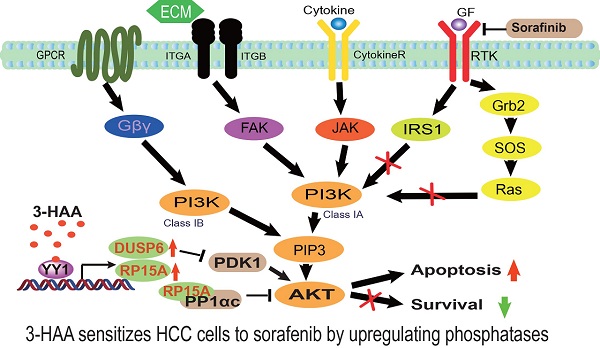
Objectives: Sorafenib is the only FDA-approved first-line target drug for HCC patients. However, sorafenib merely confers 3-5 months of survival benefit with less than 30% of HCC patients sensitive to sorafenib therapy. Thus, it's necessary to develop a sensitizer for hepatocellular carcinoma (HCC) to sorafenib. Methods: The principal component analysis, gene ontology, and KEGG analysis are utilized following RNA-sequencing. The mass spectrometry analysis following immunoprecipitation is performed to discover the phosphatase targets. Most importantly, both the cell line-derived xenograft (CDX) and the patient-derived xenograft (PDX) mouse model are used to determine the effect of 3-HAA on sorafenib-resistant HCC in vivo. Results: In nude mice carrying HCC xenograft, tumor growth is inhibited by sorafenib or 3-HAA alone. When used in combination, the treatment particularly prevents the xenograft from growing. Combined treatment also suppresses the growth of sorafenib-resistant (≥30mg/kg) PDXs. In a set of mechanistic experiments, we find enhanced AKT activation and decreased apoptotic cells in de novo and acquired sorafenib-resistant HCC cells and tissues. 3-HAA decreases AKT phosphorylation and increases the apoptosis of HCC in both cultured cells and mouse xenografts by upregulation of phosphatases PPP1R15A/DUSP6. PPP1R15A/PPP1α directly reduces Akt phosphorylation while DUSP6 decreases Akt activity through inhibiting PDK1. The AKT activator abolishes 3-HAA inhibition of HCC growth in vitro and in mice. Conclusion: This study demonstrates that 3-HAA sensitizes HCC cells to sorafenib by upregulation of phosphatases, suggesting it as a promising molecule for HCC therapy.
Keywords: 3-hydroxyanthranilc acid, sorafenib resistance, AKT, DUSP6, PPP1R15A.
 Global reach, higher impact
Global reach, higher impact