13.3
Impact Factor
Theranostics 2020; 10(12):5565-5577. doi:10.7150/thno.43569 This issue Cite
Research Paper
Chitosan modified Fe3O4/KGN self-assembled nanoprobes for osteochondral MR diagnose and regeneration
1. Institute of Nano Biomedicine and Engineering, Shanghai Engineering Research Centre for Intelligent Diagnosis and Treatment Instrument, Department of Instrument Science and Engineering, School of Electronic Information and Electrical Engineering, Shanghai Jiao Tong University, 800 Dongchuan RD, Shanghai 200240, PR China
2. Institute of Nano Biomedicine, National Engineering Center for Nanotechnology, 28 Jianchuan East RD, Shanghai 200241, PR China
3. Department of Joint Surgery and Sports Medicine, Changzheng Hospital, Second Military Medical University, 415 Fengyang RD, Shanghai 200003, PR China
4. Department of Radiology, Ren Ji Hospital, School of Medicine, Shanghai Jiao Tong University, 160 Pujian RD, Shanghai 200127, PR China
5. School of Computer Science, Faculty of Engineering, University of Sydney, NSW 2006, Australia
6. Murray Maxwell Biomechanics Laboratory, Kolling Institute, Royal North Shore Hospital, University of Sydney, NSW 2065, Australia
Abstract
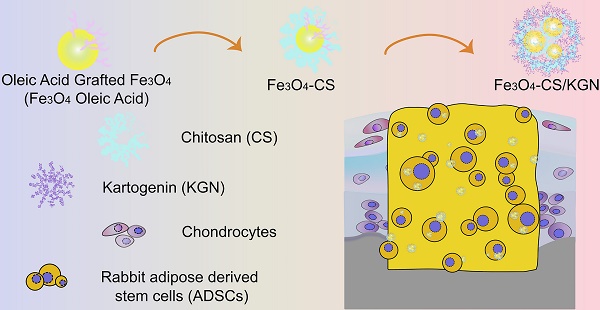
Chondral and osteochondral defects caused by trauma or pathological changes, commonly progress into total joint degradation, even resulting in disability. The cartilage restoration is a great challenge because of its avascularity and limited proliferative ability. Additionally, precise diagnosis using non-invasive detection techniques is challenging, which increases problems associated with chondral disease treatment.
Methods: To achieve a theranostic goal, we used an integrated strategy that relies on exploiting a multifunctional nanoprobe based on chitosan-modified Fe3O4 nanoparticles, which spontaneously self-assemble with the oppositely charged small molecule growth factor, kartogenin (KGN). This nanoprobe was used to obtain distinctively brighter T2-weighted magnetic resonance (MR) imaging, allowing its use as a positive contrast agent, and could be applied to obtain accurate diagnosis and osteochondral regeneration therapy.
Results: This nanoprobe was first investigated using adipose tissue-derived stem cells (ADSCs), and was found to be a novel positive contrast agent that also plays a significant role in stimulating ADSCs differentiation into chondrocytes. This self-assembled probe was not only biocompatible both in vitro and in vivo, contributing to cellular internalization, but was also used to successfully make distinction of normal/damaged tissue in T2-weighted MR imaging. This novel combination was systematically shown to be biosafe via the decrement of apparent MR signals and elimination of ferroferric oxide over a 12-week regeneration period.
Conclusion: Here, we established a novel method for osteochondral disease diagnosis and reconstruction. Using the Fe3O4-CS/KGN nanoprobe, it is easy to distinguish the defect position, and it could act as a tool for dynamic observation as well as a stem cell-based therapy for directionally chondral differentiation.
Keywords: Fe3O4-CS/KGN nanoprobe, self-assembly, theranostic strategy, MRI, osteochondral regeneration therapy
 Global reach, higher impact
Global reach, higher impact