13.3
Impact Factor
Theranostics 2020; 10(12):5181-5194. doi:10.7150/thno.44693 This issue Cite
Research Paper
A lab-on-a-disc platform enables serial monitoring of individual CTCs associated with tumor progression during EGFR-targeted therapy for patients with NSCLC
1. Center for Soft and Living Matter, Institute for Basic Science (IBS), Ulsan 44919, Republic of Korea
2. Department of Biomedical Engineering, School of Life Sciences, Ulsan National Institute of Science and Technology (UNIST), Ulsan 44919, Republic of Korea
3. Department of Pathology, Stanford University, Stanford, CA, 94305, USA
4. Clinomics Inc., Ulsan 44919, Republic of Korea
5. Department of Internal Medicine, Pusan National University School of Medicine and Biomedical Research Institute, Pusan National University Hospital, 179, Gudeok-ro, Seo-Gu, Busan 49241, Republic of Korea
#Alarice C. Lowe's previous affiliation: Brigham and Women's Hospital, Harvard Medical School, Boston, MA, USA
*These authors contributed equally to this work.
Abstract
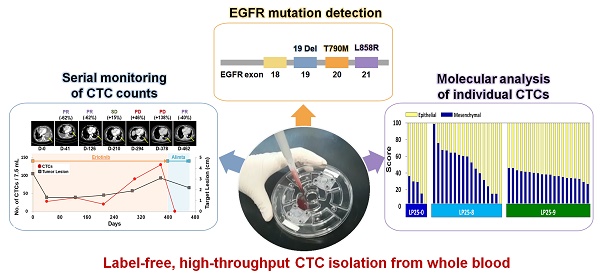
Rationale: Unlike traditional biopsy, liquid biopsy, which is a largely non-invasive diagnostic and monitoring tool, can be performed more frequently to better track tumors and mutations over time and to validate the efficiency of a cancer treatment. Circulating tumor cells (CTCs) are considered promising liquid biopsy biomarkers; however, their use in clinical settings is limited by high costs and a low throughput of standard platforms for CTC enumeration and analysis. In this study, we used a label-free, high-throughput method for CTC isolation directly from whole blood of patients using a standalone, clinical setting-friendly platform.
Methods: A CTC-based liquid biopsy approach was used to examine the efficacy of therapy and emergent drug resistance via longitudinal monitoring of CTC counts, DNA mutations, and single-cell-level gene expression in a prospective cohort of 40 patients with epidermal growth factor receptor (EGFR)-mutant non-small cell lung cancer.
Results: The change ratio of the CTC counts was associated with tumor response, detected by CT scan, while the baseline CTC counts did not show association with progression-free survival or overall survival. We achieved a 100% concordance rate for the detection of EGFR mutation, including emergence of T790M, between tumor tissue and CTCs. More importantly, our data revealed the importance of the analysis of the epithelial/mesenchymal signature of individual pretreatment CTCs to predict drug responsiveness in patients.
Conclusion: The fluid-assisted separation technology disc platform enables serial monitoring of CTC counts, DNA mutations, as well as unbiased molecular characterization of individual CTCs associated with tumor progression during targeted therapy.
Keywords: circulating tumor cells, single cell analysis, gene expression, EGFR mutation, non-small cell lung cancer
 Global reach, higher impact
Global reach, higher impact