Impact Factor
Theranostics 2020; 10(23):10434-10447. doi:10.7150/thno.47286 This issue Cite
Research Paper
HRG switches TNFR1-mediated cell survival to apoptosis in Hepatocellular Carcinoma
1. Guangdong Provincial Key Laboratory of Viral Hepatitis Research, Hepatology Unit and Department of Infectious Diseases, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China.
2. State Key Laboratory of Organ Failure Research, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China.
3. Department of Radiation Oncology, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China.
4. Department of Medical Quality Management, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China.
*These authors contributed equally to this work.
Abstract
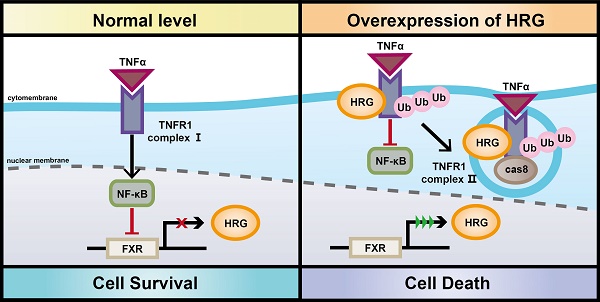
Background: Tumor necrosis factor receptor 1 (TNFR1) signaling plays a pleiotropic role in the development of hepatocellular carcinoma (HCC). The formation of TNFR1-complex I supports cell survival while TNFR1-complex II leads to apoptosis, and the underlying mechanisms of the transformation of these TNFR1 complexes in HCC remain poorly defined.
Methods: The interaction protein of TNFR1 was identified by GST pulldown assay, immunoprecipitation and mass spectrometry. In vitro and in vivo assay were performed to explore the biological features and mechanisms underlying the regulation of TNFR1 signals by histidine-rich glycoprotein (HRG). Data from the public databases and HCC samples were utilized to analyze the expression and clinical relevance of HRG.
Results: HRG directly interacted with TNFR1 and stabilized TNFR1 protein by decreasing the Lys(K)-48 ubiquitination mediated-degradation. The formation of TNFR1-complex II was prompted by HRG overexpression via upregulating Lys(K)-63 ubiquitination of TNFR1. Besides, overexpression of HRG suppressed expression of pro-survival genes by impairing the activation of NF-κB signaling in the presence of TNFR1. Moreover, downregulation of HRG was a result of feedback inhibition of NF-κB activation in HCC. In line with the pro-apoptotic switch of TNFR1 signaling after HRG induction, overexpression of HRG inhibited cell proliferation and increased apoptosis in HCC.
Conclusions: Our findings illustrate a crucial role for HRG in suppressing HCC via inclining TNFR1 to a pro-apoptotic cellular phenotype. Restoring HRG expression in HCC tissues might be a promising pharmacological approach to blocking tumor progression by shifting cellular fate from cell survival to apoptosis.
Keywords: histidine-rich glycoprotein, tumor necrosis factor receptor 1, apoptosis
 Global reach, higher impact
Global reach, higher impact