Impact Factor
Theranostics 2019; 9(23):6936-6948. doi:10.7150/thno.37380 This issue Cite
Research Paper
Lipopolysaccharide-anchored macrophages hijack tumor microtube networks for selective drug transport and augmentation of antitumor effects in orthotopic lung cancer
School of Pharmaceutical Sciences, Sun Yat-sen University, University Town, Guangzhou, 510006, P.R. China
Abstract
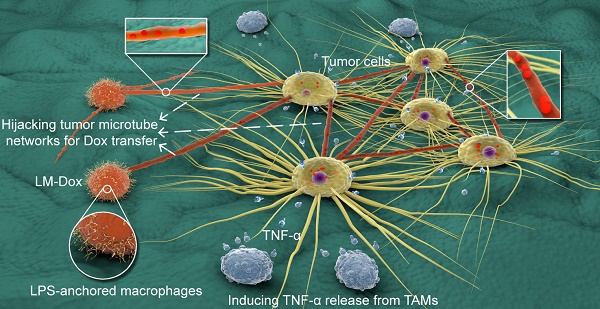
Objective: Engineered immune cells (e.g., therapeutic T cells) provide a revolutionary approach to combat cancer. Certain activated immune cells can exquisitely sense and respond to the tumor microenvironment. Here, we propose a paradigm based on engineering macrophages to allow selective intercellular drug delivery and augmentation of antitumor activities by hijacking tumor microtube networks.
Methods: Macrophages were engineered via anchoring lipopolysaccharides on the plasma membrane (LM). The tumor tropism of LM encapsulating doxorubicin (LM-Dox) was monitored by a real-time cell migration assay and small animal in vivo imaging. Monocyte chemoattractant protein-1 (CCL2) was measured by quantitative PCR and ELISA. Intercellular conduit formation was characterized by confocal laser scanning microscopy and scanning electron microscopy. LM-Dox activation of tumor-associated macrophages to release TNF-α was evaluated by western blot and immunofluorescence assays. The potential therapeutic effects of LM-Dox in a 3D tumor-immune model and a murine orthotopic lung cancer model were tested.
Results: LM-Dox exhibited tumor tropism in response to CCL2 produced by A549 lung tumor cells and lung tumor tissues resulting in a remarkably higher amount of tumor accumulation than the case of Lipo-Dox (~ 4-fold). Intriguingly, LM-Dox accumulated at tumor sites hijacked the established tumor microtube networks and even stimulated microtube formation with tumor cells but not with normal cells to enable selective and rapid transport of the drug to tumor cells. Simultaneously, LM-Dox induced secretion of TNF-α in tumor-associated macrophages, which increased the antitumor activity of Dox. Thus, LM-Dox increased the inhibitory effects on tumor growth and metastasis in a mouse orthotopic lung cancer model and minimized the side effects of Dox-induced tumor invasion.
Conclusion: Lipopolysaccharide-anchored macrophages that can hijack tumor microtube networks for selective drug transport may serve as versatile bioactive carriers of anticancer drugs. In the clinical context, these engineered microphages represent a personalized medicine approach that can be translated into potential use of patient-derived monocytes/macrophages for drug delivery by means of cell-to-cell communication.
Keywords: engineered macrophage, tumor microtube network, tumor tropism, TNF-α release, orthotopic lung cancer
 Global reach, higher impact
Global reach, higher impact