13.3
Impact Factor
Theranostics 2019; 9(12):3580-3594. doi:10.7150/thno.33141 This issue Cite
Research Paper
An in situ microenvironmental nano-regulator to inhibit the proliferation and metastasis of 4T1 tumor
1. School of Pharmaceutical Sciences, Zhengzhou University, Zhengzhou, China
2. Key Laboratory of Targeting Therapy and Diagnosis for Critical Diseases, Henan Province
3. Collaborative Innovation Center of New Drug Research and Safety Evaluation, Henan Province, Zhengzhou, China
Abstract
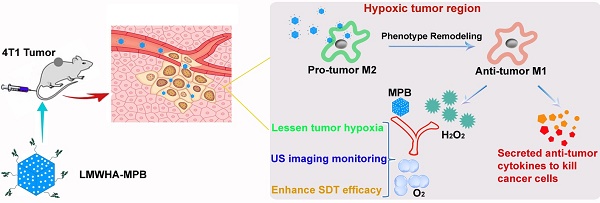
Tumor microenvironment, such as hypoxia and presence of immune cells, plays a critical role in cancer initiation, growth as well as progression, and seriously affects antitumor effect. Accordingly, we constructed a kind of multifunctional nanoparticles (NPs) with macrophage transformation and oxygen (O2) generation characteristics, to regulate the tumor microenvironment.
Methods: In this study, we synthesized mesoporous Prussian blue (MPB) NPs with low molecular weight hyaluronic acid (LMWHA) surface modification (LMWHA-MPB), and discovered that LMWHA-MPB could be used as an in situ macrophages converter and O2 generator.
Results: In vitro results showed after uptake by M2 macrophages, LMWHA-MPB displayed the potential in remodeling tumor-associated macrophages (TAMs) phenotype (pro-tumor M2→anti-tumor M1), and anti-metastatic effect on 4T1 cells. Furthermore, in vivo visualized near-infrared (NIR) imaging data proved IR783 labeled LMWHA-MPB NPs could selectively accumulate in tumor sites. Then plenty of O2 generated to alleviate tumor hypoxia via catalytic decomposition of endogenous hydrogen peroxide (H2O2). Based on these outstanding characteristics, LMWHA-MPB NPs were adopted as multifunctional nanocarriers to load sonosensitizer hematoporphyrin monomethyl ether (HMME) for O2 self-provided sonodynamic therapy (SDT). In vivo anti-tumor results showed LMWHA-MPB/HMME could effectively inhibit the proliferation and metastasis of 4T1 tumors by improving tumor microenvironment.
Conclusion: The multifunctional NPs can be used as in situ microenvironmental nano-regulators to inhibit the proliferation and metastasis of 4T1 tumor.
Keywords: mesoporous Prussian blue, low molecular weight hyaluronic acid, tumor microenvironment, SDT, tumor associated macrophages
 Global reach, higher impact
Global reach, higher impact