13.3
Impact Factor
Theranostics 2019; 9(10):2739-2753. doi:10.7150/thno.33452 This issue Cite
Research Paper
Effective targeted therapy for drug-resistant infection by ICAM-1 antibody-conjugated TPGS modified β-Ga2O3:Cr3+ nanoparticles
1. College of Pharmaceutical Sciences, Zhejiang University, 866 Yuhangtang Road, Hangzhou 310058, China.
2. Department of pharmacy, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, 310006, China.
3. State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, Collaborative Innovation Center for Diagnosis and Treatment of Infectious Diseases, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, 310006, China.
Abstract
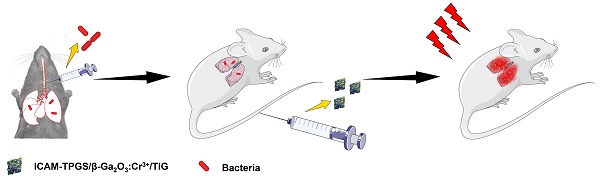
The prevalence of antibiotic resistance and lack of alternative drugs have posed an increasing threat to public health. Here, we prepared β-Ga2O3:Cr3+ nanoparticles modified with ICAM1-antibody-conjugated TPGS (I-TPGS/Ga2O3) as a novel antibiotic carrier for the treatment of drug-resistant infections.
Methods: I-TPGS/Ga2O3 were firstly characterized by measuring particle size, morphology, crystal structure, drug loading capacity, and in vitro drug release behaviors. The in vitro antibacterial activities of I-TPGS/Ga2O3/TIG were evaluated using standard and drug-resistant bacteria. The internalization of I-TPGS/Ga2O3 was observed by fluorescence confocal imaging, and the expression levels of the efflux pump genes of TRKP were analyzed by real-time RT-PCR. In vitro cellular uptake and in vivo biodistribution study were performed to investigate the targeting specificity of I-TPGS/Ga2O3 using HUEVC and acute pneumonia mice, respectively. The in vivo anti-infective efficacy and biosafety of I-TPGS/Ga2O3/TIG were finally evaluated using acute pneumonia mice.
Results: It was found that TPGS could down-regulate the over-expression of the efflux pump genes, thus decreasing the efflux pump activity of bacteria. I-TPGS/Ga2O3 with small particle size and uniform distribution facilitated their internalization in bacteria, and the TPGS modification resulted in a significant reduction in the efflux of loaded antibiotics. These properties rendered the encapsulated tigecycline to exert a stronger antibacterial activity both in vitro and in vivo. Additionally, targeted delivery of I-TPGS/Ga2O3 mediated by ICAM1 antibodies contributed to a safe and effective therapy.
Conclusion: It is of great value to apply I-TPGS/Ga2O3 as a novel and effective antibiotic delivery system for the treatment of drug-resistant infections.
Keywords: β-Ga2O3:Cr3+, Tocopherol polyethylene glycol succinate, Drug-resistance bacteria, Targeted antibiotic delivery, Bioimaging
 Global reach, higher impact
Global reach, higher impact