13.3
Impact Factor
Theranostics 2018; 8(9):2377-2386. doi:10.7150/thno.23567 This issue Cite
Research Paper
A Supramolecular Approach for Liver Radioembolization
1. Interventional Molecular Imaging Laboratory, Department of Radiology, Leiden University Medical Center, Leiden, The Netherlands.
2. Department of Agrotechnology and Food services, subdivision BioNanoTechnology, Wageningen University, Wageningen, The Netherlands
3. Department of Radiology, Nuclear Medicine Section, Leiden University Medical Center, Leiden The Netherlands.
4. Interventional Radiology, Department of Radiology, Leiden University Medical Center, Leiden, The Netherlands
5. Molecular NanoFabrication group, MESA+ Institute for Nanotechnology, University of Twente, Enschede, The Netherlands
*These authors contributed equally to this work
Abstract
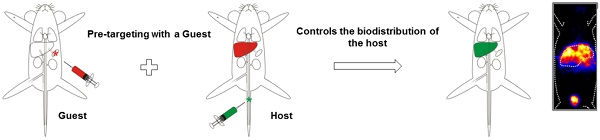
Hepatic radioembolization therapies can suffer from discrepancies between diagnostic planning (scout-scan) and the therapeutic delivery itself, resulting in unwanted side-effects such as pulmonary shunting. We reasoned that a nanotechnology-based pre-targeting strategy could help overcome this shortcoming by directly linking pre-interventional diagnostics to the local delivery of therapy.
Methods: The host-guest interaction between adamantane and cyclodextrin was employed in an in vivo pre-targeting set-up. Adamantane (guest)-functionalized macro albumin aggregates (MAA-Ad; d = 18 μm) and (radiolabeled) Cy5 and β-cyclodextrin (host)-containing PIBMA polymers (99mTc-Cy50.5CD10PIBMA39; MW ~ 18.8 kDa) functioned as the reactive pair. Following liver or lung embolization with (99mTc)-MAA-Ad or (99mTc)-MAA (control), the utility of the pre-targeting concept was evaluated after intravenous administration of 99mTc-Cy50.5CD10PIBMA39.
Results: Interactions between MAA-Ad and Cy50.5CD10PIBMA39 could be monitored in solution using confocal microscopy and were quantified by radioisotope-based binding experiments. In vivo the accumulation of the MAA-Ad particles in the liver or lungs yielded an approximate ten-fold increase in accumulation of 99mTc-Cy50.5CD10PIBMA39 in these organs (16.2 %ID/g and 10.5 %ID/g, respectively) compared to the control. Pre-targeting with MAA alone was shown to be only half as efficient. Uniquely, for the first time, this data demonstrates that the formation of supramolecular interactions between cyclodextrin and adamantane can be used to drive complex formation in the chemically challenging in vivo environment.
Conclusion: The in vivo distribution pattern of the cyclodextrin host could be guided by the pre-administration of the adamantane guest, thereby creating a direct link between the scout-scan (MAA-Ad) and delivery of therapy.
Keywords: Nanotechnology, Interventional radiology, Pre-targeting, Supramolecular chemistry, Radioembolization
 Global reach, higher impact
Global reach, higher impact