Impact Factor
Theranostics 2017; 7(19):4643-4657. doi:10.7150/thno.20418 This issue Cite
Research Paper
Detection of Treatment Success after Photodynamic Therapy Using Dynamic Contrast-Enhanced Magnetic Resonance Imaging
1. Eindhoven University of Technology, Biomedical Engineering Department, Biomedical NMR, Eindhoven, The Netherlands
2. Philips Research, Oncology Solutions, Eindhoven, The Netherlands
3. University Medical Center Utrecht, Department of Radiology, Utrecht, The Netherlands
4. Academic Medical Center, Biomedical Engineering and Physics, Amsterdam, The Netherlands
†This work was dedicated to Professor Klaas Nicolay, our friend, mentor, and a devoted researcher, who passed away on January 10, 2017. We will remember him for his great contribution to science and education in the field of Biomedical NMR, and his warm and inspiring personality.
Abstract
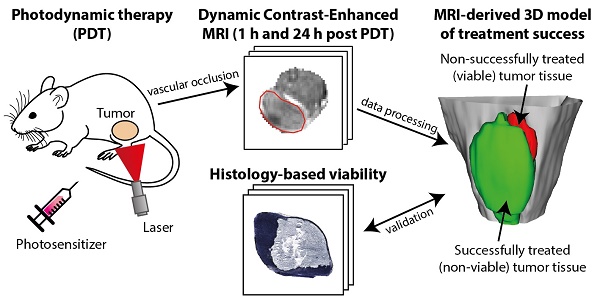
Early evaluation of response to therapy is crucial for selecting the optimal therapeutic follow-up strategy for cancer patients. PDT is a photochemistry-based treatment modality that induces tumor tissue damage by cytotoxic oxygen radicals, generated by a pre-injected photosensitive drug upon light irradiation of tumor tissue. Vascular shutdown is an important mechanism of tumor destruction for most PDT protocols. In this study, we assessed the suitability of Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) to evaluate treatment efficacy within a day after photodynamic therapy (PDT), using the tumor vascular response as a biomarker for treatment success.
Methods: DCE-MRI at 7 T was used to measure the micro-vascular status of subcutaneous colon carcinoma tumors before, right after, and 24 h after PDT in mice. Maps of the area under the curve (AUC) of the contrast agent concentration were calculated from the DCE-MRI data. Besides, tracer kinetic parameters including Ktrans were calculated using the standard Tofts-Kermode model. Viability of tumor tissue at 24 h after PDT was assessed by histological analysis.
Results: PDT led to drastic decreases in AUC and Ktrans or complete loss of enhancement immediately after treatment, indicating a vascular shutdown in treated tumor regions. Histological analysis demonstrated that the treatment induced extensive necrosis in the tumors. For PDT-treated tumors, the viable tumor fraction showed a strong correlation (ρ ≥ 0.85) with the tumor fraction with Ktrans > 0.05 min-1 right after PDT. The viable tumor fraction also correlated strongly with the enhanced fraction, the average Ktrans, and the fraction with Ktrans > 0.05 min-1 at 24 h after PDT. Images of the viability stained tumor sections were registered to the DCE-MRI data, demonstrating a good spatial agreement between regions with Ktrans > 0.05 min-1 and viable tissue regions. Finally, 3D post-treatment viability detection maps were constructed for the tumors of three mice by applying a threshold (0.05 min-1) to Ktrans at 24 h after PDT. As a proof of principle, these maps were compared to actual tumor progression after one week. Complete tumor response was correctly assessed in one animal, while residual viable tumor tissue was detected in the other two at the locations where residual tumor tissue was observed after one week.
Conclusion: This study demonstrates that DCE-MRI is an effective tool for early evaluation of PDT tumor treatment.
Keywords: Dynamic contrast-enhanced magnetic resonance imaging, treatment response evaluation, mouse tumor model, photodynamic therapy, tumor perfusion
 Global reach, higher impact
Global reach, higher impact