13.3
Impact Factor
Theranostics 2014; 4(1):106-118. doi:10.7150/thno.7409 This issue Cite
Research Paper
uPAR-targeted Optical Imaging Contrasts as Theranostic Agents for Tumor Margin Detection
1. Department of Surgery, Emory University School of Medicine, 1365-C Clifton Road, Atlanta, GA, USA.
2. Department of Radiology and Imaging Sciences, Emory University School of Medicine, 1364-C Clifton Road, Atlanta, GA, USA.
3. Department of Hematology and Oncology, Emory University School of Medicine, 1365-C Clifton Road, Atlanta, GA, USA.
4. Ocean Nanotech, LLC, Springdale, AR, USA.
Abstract
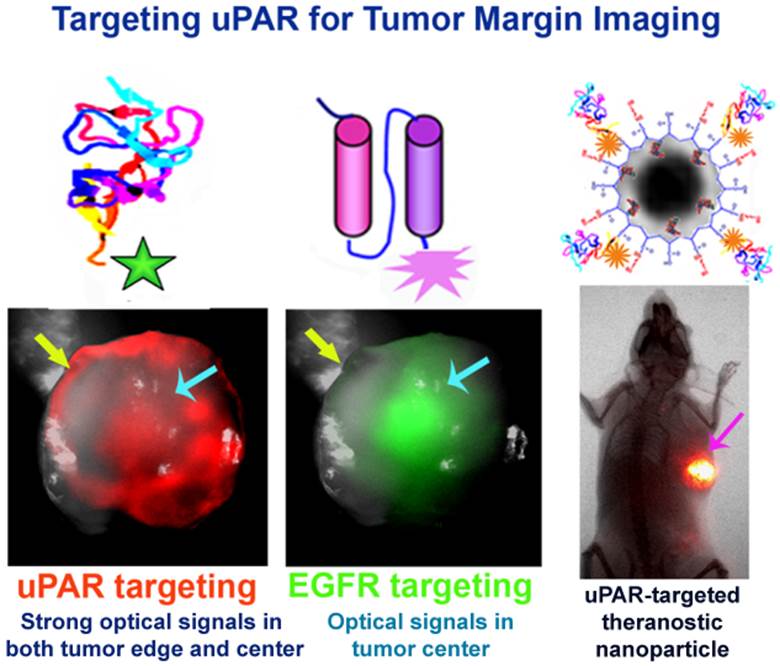
Complete removal of tumors by surgery is the most important prognostic factor for cancer patients with the early stage cancers. The ability to identify invasive tumor edges of the primary tumor, locally invaded small tumor lesions, and drug resistant residual tumors following neoadjuvant therapy during surgery should significantly reduce the incidence of local tumor recurrence and improve survival of cancer patients. In this study, we report that urokinase plasminogen activator (uPA) and its receptor (uPAR) are the ligand/cell surface target pair for the development of targeted optical imaging probes for enhancing imaging contrasts in the tumor border. Recombinant peptides of the amino terminal fragment (ATF) of the receptor binding domain of uPA were labeled with near infrared fluorescence (NIR) dye molecules either as peptide-imaging or peptide-conjugated nanoparticle imaging probes. Systemic delivery of the uPAR-targeted imaging probes in mice bearing orthotopic human breast or pancreatic tumor xenografts or mouse mammary tumors led to the accumulation of the probes in the tumor and stromal cells, resulting in strong signals for optical imaging of tumors and identification of tumor margins. Histological analysis showed that a high level of uPAR-targeted nanoparticles was present in the tumor edge or active tumor stroma immediately adjacent to the tumor cells. Furthermore, following targeted therapy using uPAR-targeted theranostic nanoparticles, residual tumors were detectable by optical imaging through the imaging contrasts produced by NIR-dye-labeled theranostic nanoparticles in drug resistant tumor cells. Therefore, results of our study support the potential of the development of uPAR-targeted imaging and theranostic agents for image-guided surgery.
Keywords: uPAR, optical imaging, theranostic nanoparticles, tumor margin, and image-guided surgery
 Global reach, higher impact
Global reach, higher impact