Impact Factor
Theranostics 2025; 15(11):5045-5072. doi:10.7150/thno.106465 This issue Cite
Review
Sex bias in tumor immunity: insights from immune cells
1. School of Medicine, University of Electronic Science and Technology of China, Chengdu, China.
2. Department of Radiation Oncology, Radiation Oncology Key Laboratory of Sichuan Province, Sichuan Clinical Research Center for Cancer, Sichuan Cancer Hospital & Institute, Sichuan Cancer Center, Affiliated Cancer Hospital of University of Electronic Science and Technology of China, Chengdu, China.
3. Department of Urology, Sichuan Provincial People's Hospital, School of Medicine, University of Electronic Science and Technology of China, Chengdu, China.
4. Department of Nephrology, Sichuan Provincial People's Hospital, School of Medicine, University of Electronic Science and Technology of China, Chengdu, China.
5. Department of Organ Transplantation, Sichuan Provincial People's Hospital, School of Medicine, University of Electronic Science and Technology of China, Chengdu, China.
† These authors contributed equally to this work.
Received 2024-11-6; Accepted 2025-3-8; Published 2025-3-31
Abstract
Significant sex disparities have been observed in cancer incidence, treatment response to immunotherapy, and susceptibility to adverse effects, affecting both reproductive and non-reproductive organ cancers. While lifestyle factors, carcinogenic exposure, and healthcare access contribute to these disparities, they do not fully explain the observed male-female variation in anti-tumor immunity. Despite the preferential expression of sex hormone receptors in immune cells, X chromosome also contains numerous genes involved in immune function, and its incomplete inactivation may enhance anti-tumor immune responses in females. In contrast, loss or downregulation of Y-linked genes in males has been associated with an increased cancer risk. Additionally, estrogen, progesterone and androgen signaling pathways influence both innate and adaptive immune responses, contributing to sex-specific outcomes in cancer progression and therapy. Sex-biased differences are also evident in the epigenetic regulation of gene expression, cellular senescence, microbiota composition, metabolism, and DNA damage response, all of which impact anti-tumor immunity and immunotherapy treatment efficacy. In general, the combination of sex chromosomes, sex hormones, and hormone receptors orchestrates the phenotype and function of various immune cells involved in tumor immunity. However, sex disparity in each specific immune cell are context and environment dependent, considering the preferential expression of hormone receptor in immune cell and sex hormone levels fluctuate significantly across different life stages. This review aims to outline the molecular, cellular, and epigenetic changes in T cells, B cells, NK cells, DCs, neutrophils, and macrophages driven by sex chromosomes and sex hormone signaling. These insights may inform the design of sex-specific targeted therapies and leading to more individualized cancer treatment strategies.
Keywords: Tumor immunity, Sex bias, Tumor microenvironment, Sex hormones, Immunotherapy
1. Background
Although men and women share nearly identical genomes, they exhibit distinct susceptibilities to cancer and differences in prognosis. The sex bias is observed not only in sex-specific cancers like breast and prostate cancer, but also in cancers originating from non-reproductive organs [1]. In general, male patients experience higher mortality rates and shorter survival compared to females [2]. While social behaviors, environmental exposures, and access to medical care contribute to these disparities, they account for only a fraction of the overall cancer risk [3]. A prospective cohort study on 21 cancer types found that men have a higher risk of developing cancer at most shared anatomic sites compared to women. This increased risk persists even after accounting for behavioral risk factors and carcinogenic exposures, suggesting that sex-related biological factors including sex-biased immunity, may play a key role [3].
Sex-biased immunity has been well established both in innate and adaptive immunity, which have been observed in conditions such as inflammatory diseases [4], organ transplantation [5-7], systemic lupus erythematosus (SLE), various autoimmune disorders [8], as well as in COVID-19 [9] and ischemic stroke [10]. These differences also influence tumor immunity, which constantly participated in the process of oncogenesis, cancer progression, treatment responses and immune-related side effects. Factors contributing to cancer immune variability between genders include sex hormones, sex chromosomes [11, 12].
Tumor immunity can be affected by sex hormones, especially estrogen and androgen though its receptors in innate and adaptive immune cells. Estrogen signaling is primarily driven by the intracellular estrogen receptor (ER)-estrogen response element (ERE) complex [13, 14], which forms when ER bind to the ERE on DNA, either as homodimers or heterodimers [15, 16]. The binding inevitably leads to the rapid activation of several signaling pathways, including the extracellular signal-regulated kinase (ERK)/mitogen-activated protein kinase (MAPK) pathway, cAMP-regulated gene transcription, protein kinase C (PKC)-mediated increases in intracellular Ca2+ levels, and JNK/phosphatidylinositol 3 kinase (PI3K) pathways, which regulate apoptosis [17]. Classical membrane-bound 17β-estradiol (E2)-ER binding triggers multiple rapid signaling cascades [18, 19]. Androgen signaling begins when the androgen receptor (AR) binds to its ligand, dihydrotestosterone (DHT), triggering a conformational change. This causes the AR to dissociate from heat shock proteins and enter the nucleus, where it undergoes phosphorylation and dimerization. The activated AR then binds to androgen response elements (AREs) in the promoters of target genes, recruiting co-activators to form transcriptional complexes. This process ultimately drives the transcription of genes that promote cancer progression and invasion [17, 20]. However, sex hormones can ignite anti-tumor immunity and induce pro-tumor immunity at the same time through signaling on various immune cells, e.g., ER signaling pathways enhance the anti-tumor effects of most immune cells, while concurrently impairing macrophage-mediated tumor immunity in endometrial cancer, leading to the complexity of understanding the roles of each hormone.
Factors contributing to tumor immune responses include sex chromosomes and subsequent epigenetic modifications and coding genes in different immune cells as well. X-chromosomes are equipped with more immune functional genes including TLR7, FOXP3, and CD40L, and epigenetic modifying genes such as KDM6A, while X chromosome inactivation (XCI) upregulates or impairs anti-tumor immunity of various immune cells [21]. Y chromosome genes, once thought to primarily regulate autoimmune diseases [22], may also impact cancer risk. Y chromosome mosaic loss (LOY) has been linked to an increase in regulatory T cell (Treg) numbers and activity, potentially heightening cancer susceptibility [23].
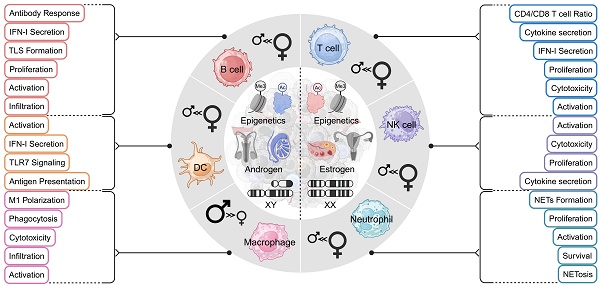
In general, both innate and adaptive immunity tend to be stronger in women, though the impact of sex hormone signaling and chromosomal differences on specific immune cells can be context dependent. For instance, tumor antigen presentation by dendritic cells (DCs) is enhanced in females through E2 and estrogen receptor alpha (ERα) signaling, which upregulates MHC class II expression and leads to more robust anti-tumor T cell responses. Additionally, macrophage polarization toward the M1 phenotype is promoted by AR signaling, which plays a role in immune response modulation [24, 25]. In contrast, male T cells lack certain X-linked cytotoxic genes, and AR signaling in males promotes CD8+ T cell exhaustion, fostering immune tolerance within tumors. Estrogen has been shown to facilitate anti-tumor Th1 differentiation and encourage the differentiation of tolerogenic Tregs in CD4+ T cells [12]. Of note, estrogen receptor beta (ERβ) signaling enhances CD8+ T cell activation and cytokine production through T cell receptor (TCR) stimulation, ultimately reducing tumor burden [26-28].
Improved understanding of sex biases in different immune cell may provide the insights for personalized patient management and treatment of cancer. In this context, we discuss the sex-biased molecular, cellular, and epigenetic regulation of anti-tumor immunity in various immune cells, shaped by a combination of hormonal and chromosomal factors.
2. Sex bias in T cell tumor immunity
T cells in the tumor microenvironment (TME) are heavily influenced by cancer-related factors. Previous research has focused on the anti-tumor mechanisms of CD4+ T cells and the two-step dysfunction of CD8+ T cells induced by the TME [29, 30]. Recently, studies have identified the TME can induce a form of multi-reprogramming in T cells, which may contribute to their dysfunction. This finding offers new insights into cancer immunology and therapeutic strategies [30-38]. However, there remains a lack of research on how the TME influences pro-T cell selection and maturation. Understanding these processes could provide valuable clinical insights and pave the way for new cancer immunotherapy.
Numerous studies have demonstrated that sex hormones influence T cell function, including T-cell development, gene expression regulating and epigenetic and metabolic reprogramming, through sex hormone receptors and their downstream signaling pathways [39-41]. Specifically, progesterone (P4) and human chorionic gonadotropin (hCG) influence T cell epigenetic and metabolic reprogramming by upregulating progesterone-induced blocking factor (PIBF) and promoting histone methylation [42-44]. This leads to an overall Th2 bias and an increased proportion of Tregs, which negatively impact tumor immunity [44-46]. Recent research also identifies hCG as an autocrine tumor growth factor, and the expression of the progesterone receptor (PR) has been shown to impair the sensitivity of tumors to anti-LAG3 immunotherapy [47, 48].
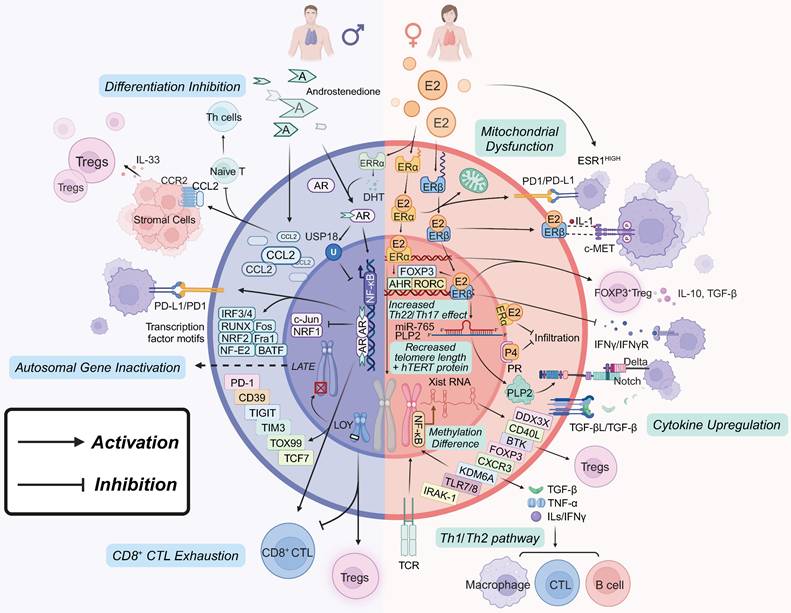
The concept of sex hormone regulated tumor responses is widely accepted, although the effects of androgens remain less well understood [49]. Furthermore, there are significant sex-based differences in the differentiation of CD4+ and CD8+ T cells. Mechanisms such as X-chromosome gene linkage, Xist-mediated XCI evasion [50], Y-chromosome loss [23], and epigenetic inheritance [51] have all been implicated in modulating T cell function. Sex bias dependent T-cell exhaustion However, non-receptor-mediated pathways through which sex hormones affect T cells still require further exploration (Figure 1).
Sex Factors in Regulating T Cell Tumor Immunity. Estrogen effects: ERα Pathway: Promotes Th2 cell differentiation and proliferation by upregulating IL-4, IL-5, IL-6, IL-10, and IL-13, while concurrently suppressing Th1 responses. It enhances the proliferation of FOXP3+ Tregs and the expression of RORC, AHR, FOXP3, and PD-1, contributing to immune tolerance and promoting Th17/Th22 infiltration. High ERα expression is associated with reduced CTLs infiltration and mitochondrial dysfunction. ERβ Pathway: Modulates T cell function through the ERβ/c-MET and ERβ/IL-1/c-MET pathways, particularly in bladder cancer. The ERβ/miR-765/PLP2/Notch axis plays a significant role in the development of endometrial cancer and mediates immune escape in NSCLC. ERβ signaling also enhances Tregs differentiation and the secretion of IL-10 and TGF-β. Androgen effects: AR signaling suppresses T cell activation and differentiation by inducing USP18 and inhibiting NF-κB, leading to increased checkpoint molecule expression and subsequent CTLs exhaustion. In VAT, androgens may function independently of AR to promote IL-33 expression and Tregs recruitment, while also inhibiting T helper cells differentiation. Sex chromosomes and epigenetic effects: Evasion of XCI impacts T cell function by influencing genes involved in Th1/Th2 pathways and promoting FOXP3+ Tregs. LOY is associated with increased numbers and activity of Tregs, reduced CTLs exhaustion, and altered T cell gene expression, all of which influence cancer susceptibility. Sex hormones influence DNA methylation and histone modifications, affecting T cell functions, cancer progression and the enrichment of certain transcription factor motifs, while decreasing others, promoting Tregs' immunosuppressive capacity.
2.1 Estrogen regulation of T cells
Estrogen influences CD4+ T cells primarily through ERs in the TME, which enhance T helper (Th) 1 and Th2 immune responses and modulate cytokine and transcription factor secretion. Notably, E2 reduces the Th1/Th2 ratio, promotes Th22 and Th17 cell infiltration, and increases Th2-type cytokines, such as IL-4, IL-5, IL-6, IL-10, and IL-13, with IL-22 being particularly elevated [52]. Elevated levels of E2 in the TME further enhance Th2 responses by downregulating IRF1 and increasing IL-4 production, thereby inhibiting Th1 activity and promoting Th2 differentiation and proliferation [53]. E2 also enhances Th22 cell activity through the AHR/ARNT/NF-κB/ERK pathway and the RORC/OREs pathway, which upregulate AHR and RORC levels. Additionally, the HER2 gene has been identified as a critical regulator of peripheral Th22 cell differentiation, working in tandem with Th17 cells to promote tumor growth [52].
Research into ERα and ERβ signaling in T-cell tumor immunity and targeted cancer therapy has provided valuable insights, but the full scope of the sex hormone signaling network remains incompletely understood. E2 promotes the proliferation and infiltration of FOXP3+ Tregs and induces the expression of programmed death-1 (PD-1) on their surface in an ER-dependent manner. This process is also thought to be mediated by GPER, which contributes to tumor immune tolerance [39, 54]. ERα signaling enhances Tregs infiltration into the TME and upregulates the expression of immune-suppressive molecules like TGF-β and IL-10 by directly interacting with the FOXP3 promoter, thereby inhibiting effector T cell activation [55]. Similar effects have been observed in non-small cell lung cancer (NSCLC), where increased ERα and PR gene expression correlates with reduced infiltration of CD4+ and CD8+ cytotoxic T lymphocytes (CTLs) [56, 57]. ERα, as a regulatory binding protein (RBP), has also been shown to reduce telomere length, mitochondrial DNA copy number, and hTERT protein expression, thus inhibiting CTLs proliferation [58]. Elevated ESR1 gene expression has been linked to upregulation of immune checkpoint genes [59]. Alternatively, ERα signaling may induce mitochondrial dysfunction in T cells [60]. Additionally, estrogen can be converted to dihydrotestosterone (DHT) via ERRα, further activating downstream signaling pathways [61].
In contrast, ERβ is primarily expressed on immune cells rather than cancer cells, where it plays a complex and generally negative role in cancer immunology. It has been shown to decrease CD8+ CTLs infiltration and cytokine release, while promoting Tregs differentiation. In bladder cancer, CD4+ T cells facilitate tumor invasion through the ERβ/c-MET and ERβ/IL-1/c-MET pathways. Inhibition of IL-1 or the use of the ERβ antagonist PHTTP can reduce the malignancy of bladder cancer [62]. Mutations in ERβ lead to increased infiltration of T cells and neutrophils in the TME [63], along with decreased interferon gamma (IFN-γ) levels, which promote tumor growth and progression [64]. ERβ signaling in CD8+ T cells can activate these cells, enhancing the efficacy of αPD-1 tumor therapy by modulating tumor-derived phosphotyrosine [27]. In prostate cancer, the estrogen/ERβ pathway can be inhibited to promote tumor invasion [65, 66]. Moreover, miR-765 from CD45RO-CD8+ T cells can limit estrogen-driven endometrial cancer development through the ERβ/miR-765/PLP2/Notch axis [26]. E2 can also impair cytotoxic of CTLs and facilitate immune escape in NSCLC cells via the ERβ/SIRT1/FOXO3a/PD-L1 axis [67]. Furthermore, ERβ signaling promotes Tregs differentiation and induces their secretion of IL-10 and TGF-β, reducing CD8+ T cell cytotoxicity and overall tumor immunoreactivity [68, 69]. Blocking ERβ-specific signaling can impair E2-mediated differentiation and function of intestinal Tregs, disrupting immune tolerance, as seen in female patients with chronic inflammatory bowel disease [70].
As the negative role of estrogen in various diseases becomes increasingly recognized, and with advancements in high-throughput technologies, platforms for discovering anti-estrogenic drugs have been developed. These platforms, combined with computational simulation techniques like molecular dynamics, have enabled the identification and efficacy prediction of estrogen and its receptor targets, as well as the development of novel therapeutic agents [71-77]. For example, the use of the ERβ agonist S-oxynivalenol in combination with αPD-1 immunotherapy has shown promise in enhancing tumor-infiltrating CD8+ T cells. Moreover, improving the outcomes of immune checkpoint inhibitor therapy (ICT) may involve counteracting the potential downregulation of PD-1/PD-L1 expression by ERα through the IL-17 signaling axis [27], thus promoting CD8+ T cell infiltration [78]. In summary, targeted therapies that address metabolic, epigenetic, and cancer-related pathways highlight the complex role of sex hormones in cancer progression and treatment [79].
2.2 Androgen regulation of T cells
Androgens play a crucial role in shaping T cell phenotypes, primarily through the AR signaling pathways. ARs in T cells are classified into intracellular androgen receptors (iAR) and membrane androgen receptors (mAR), both of which regulate T cell development, differentiation, and immune response [80-82]. The iARs, also known as cytosolic ARs, interact with key signaling pathways, including PI3K, Src family kinases, RAS GTPases, and the MAPK/ERK pathway, where it regulates cell proliferation and survival. In contrast, the non-classical mARs interact with ERK1/2 pathways and G proteins, showing a different androgen affinity compared to iARs [83]. However, distinct androgen receptor signaling functions have not yet been identified in T cells or other immune cells. In detail, androgen and AR signaling can inhibit NF-κB activation, which in turn suppresses T-cell development, maturation, Th1 differentiation, and proliferation [84-86]. Additionally, androgens can modulate the expansion and function of Tregs by increasing the expression of FOXP3 [87]. These processes impair tumor immunity and help prevent autoimmune diseases in males by promoting immune tolerance through an AREs-dependent mechanism [84, 88].
Recent advancements in tumor immunotherapy, including androgen deprivation therapy (ADT), have rekindled interest in the effects of androgens and AR signaling on T cell-mediated tumor immunity. ADT, commonly used in prostate cancer treatment, has been recognized for its ability to block AR signaling in T cells, thereby restoring T cell activation by inhibiting the PD-1/PD-L1 pathway [81]. This therapeutic effect suppresses Th differentiation in naïve T cells while promoting Tregs activation [64]. Androgen signaling has also been shown to contribute to cancer-related immunosuppression by suppressing T cell activity through the upregulation of USP18, which inhibits NF-κB activity [84]. Additionally, AR activity in T cells can lead to reduced IFN-γ production and resistance to tumor immunotherapy, diminishing the effectiveness of immune checkpoint inhibitors (ICIs). This downregulation may be counteracted by combining AR antagonists with PD-1 inhibitors [89].
ER signaling is integral to this process collectively. For example, aromatase gene knockout (KO) mice, which are unable to produce estrogen, do not develop prostate cancer despite elevated testosterone levels [90]. Recent studies have shown that estrogen promotes endoplasmic reticulum-associated degradation and enhances the expression of the proto-oncogene c-Myc in prostate cancer cells through AR/ER signaling, highlighting the complex mechanisms underlying sex hormone action [91]. In contrast, in visceral adipose tissue (VAT), androgens may act independently of AR in a sex-hormone-dependent manner to stimulate IL-33 expression in stromal cells via the CCL2/CCR2 axis [49, 92]. This mechanism increases Tregs recruitment and results in higher Tregs levels in males, thereby contributing to more effective restriction of VAT inflammation.
T-cell exhaustion progression, a factor that directly influences cancer progression and therapeutic response, is significantly modulated by androgens. It progresses more rapidly in male T cells than in female T cells, with males exhibiting a higher frequency of progenitor exhausted T cells (PEX; CD8+CD44+PD-1+TCF1+TIM3-). However, sex bias in terminally exhausted T cell (TEX; CD8+CD44+PD-1+TCF1-TIM3+) remains to be illustrated [93, 94]. In contrast, female tumor-infiltrating T cells show elevated frequencies of effector T cells (EFF; CD8+CD44+TCF1-TIM3-), along with a mild increase in inhibitory receptor expression and a decrease in cytokine expression [94]. The availability and expression of AR in intratumoral stem cell-like CD8+ T cells may drive differentiation toward terminal exhaustion and a PEX signature, impairing endogenous T cell stemness and function within the TME [95, 96]. Therapeutic CD8+ T cells with AR deficiency or blockade demonstrated a reduction in the terminally exhausted subset, downregulated immune checkpoint expression, and enhanced cytotoxicity and tumor immunity [89, 95]. Recent research has revealed that AR signaling plays a key role as a T cell-intrinsic regulator of CD8+ T cell exhaustion and in regulating the differentiation of stem cell-like CD8+ T cells into a terminally exhausted phenotype through transcriptional mechanisms [96, 97]. This is evidenced by the overexpression of T-cell immune checkpoints such as PD-1, CD39, TIGIT, LAG3, CTLA4 and TIM-3 infiltrating in tumors compared with orchiectomy or female group [84, 94]. Anti-PD-1 blockade is also more effective in male T cells, resulting in a significant reduction of exhausted T-cell subsets and increased proliferation compared to females. These changes may contribute to a survival benefit in males, as reported in studies [94]. The underlying mechanism may involve AR acting as a trans-activator of T cell factor (TCF) 7 and a sex-specific TCF7-centered regulon in CD8+ T cells [96, 98]. Additionally, CUT&Tag-seq analyses have shown that AR directly binds to the promoters of key transcriptional regulators of T cell exhaustion, including TCF7 and TOX [97, 99].
AR signaling also mediates epigenetic changes in T cells, resulting in increased enrichment of transcription factor motifs such as Fra1, Fos, NF-E2, RUNX, IRF3, IRF4, NRF2 (NFE2L2), and BATF, while decreasing the enrichment of c-Jun and NRF1 motifs in male 2C-TCR CD8+ T cells in antitumor immunity [97]. Meanwhile, AR pathways induced epigenetic changes can lead to metabolic and epigenetic reprogramming of T cells [100]. Androgens inhibit the expression of Delta-like 4 (Dll4), a Notch ligand essential for T cell progenitor commitment and differentiation, in a dose-dependent manner in cortical thymic epithelial cells (cTECs) [101]. This regulation occurs through androgen-response elements in the Dll4 gene promoter, where androgen/AR complexes are recruited, as demonstrated by chromatin immunoprecipitation [101]. In non-TME contexts, androgens promote self-tolerance by upregulating the expression of autoimmune regulator (Aire) in medullary thymic epithelial cells (mTECs). This occurs through direct binding to the Aire gene promoter, resulting in more efficient negative selection of self-reactive T cells [102], while estrogen decrease the expression of Aire by epigenetically regulating its methylation [103].
2.3 Sex chromosome regulation of T cells
Sex differences in T cell phenotypes can be attributed to Xist-mediated XCI evasion [50]. Key signaling pathways, such as TCR and NF-κB, play a role in maintaining these differences. Several genes that evade XCI in CD4+ and CD8+ T cells, including CD40L, CXCR3, IRAK-1, BTK, DDX3X, and TLR7/TLR8, influence Th1 and Th17 differentiation and are linked to autoimmune conditions [104]. These genes are more prevalent in females [50, 105, 106]. For example, the FOXP3 gene, which is crucial for tumor resident Tregs function, is more highly expressed in female CD8+ T cells and tumor resident T cells due to XCI evasion, contributing to decreased tumor immunoreactivity [107, 108]. The KDM6A gene, which escapes XCI and produces UTX, modulates Th1 and Th2 pathways and influences immune function in NK cells, leading to more pronounced autoimmune responses in females [39, 109]. KDM6A has also been identified as a critical target for immunotherapy in glioblastoma. In male glioblastoma patients, lower KDM6A expression compared to females is linked to higher levels of immune suppression and T-cell exhaustion, resulting in a less effective immunotherapy response [110]. The reduction in T-cell durability, influenced by UTX in the chronic inflammatory cancer microenvironment, may explain this difference [111].
Although research on the X chromosome's role in cancer remains limited, a recent review highlights the similarities in T cell behavior, including epigenetic modifications, gene expression, cytotoxicity, and especially exhaustion, in chronic inflammation and autoimmune diseases [112, 113]. Many studies have integrated the role of T cells in the three conditions to explore these areas collectively [112, 114, 115]. A clinical review also suggested that autoimmune responses may be beneficial for cancer prognosis [116]. We anticipate that the influence of sex chromosome-related factors on tumor-infiltrating T cells will be uncovered in future research, and these findings could contribute to the development of X chromosome-targeted therapies for cancer.
Recent findings also suggest that Y chromosome genes, once thought to primarily regulate autoimmune diseases [22], may also impact cancer risk. Y chromosome mosaic loss (LOY) has been linked to an increase in Tregs numbers and activity, potentially heightening cancer susceptibility [23]. T cells with LOY exhibit altered expression of autosomal genes, such as LATE [117], which can contribute to CD8+ T cell exhaustion and dysfunction, facilitating tumor escape from T cell-mediated killing [118]. Interestingly, LOY tumors have shown increased sensitivity to ICI therapy, suggesting a potential avenue for effective treatment [118].
2.4 Epigenetic regulation of T cells
Epigenetic changes governed by sex hormones and chromosomes offer promise for future treatments of "cold" tumors, particularly when combined with ICIs and adoptive T cell therapy [119]. The sex-differentiated manifestations observed in various allergic diseases and asthma are often regulated by methylation and histone modifications of relevant genes. For instance, genes such as IFNG, IL17A, and IL17F in Th cells, as well as RORC, FOXP3, and IL4 in Tregs, display high levels of H3K9me2 marking, while methylation markers in different T cell subsets exhibit considerable variation [120]. This mechanism may stem from multiple ERα-binding sites within the promoters of DNA methyltransferase isoforms DNMT1, DNMT3A, and DNMT3B, along with the sex-specific effects of the dosage compensation mechanism that inactivates the female X chromosome, impacting methylation sites on the X chromosome [121].
Moreover, epigenetic mechanisms involved in carcinogenesis within T cells have been identified. For example, the normalization of FOXP3 expression relative to Tregs numbers affects the non-oxidative pentose phosphate pathway (PPP), leading to the downregulation of Tregs' immunosuppressive capacity due to a deficiency in transketolase (TKT), an essential enzyme in the non-oxidative PPP. This epigenetic downregulation increases the risk of carcinogenesis [122]. The senescence characteristic of CD8+ TEMRA cells in human blood is also influenced by the methylation of Cytosine-phosphate-Guanine dinucleotide (CpG) motifs [123], which could open avenues for therapeutic intervention.
Recent research has introduced methods such as EPRIM [124], which evaluates the pre-efficacy of anti-ER therapy based on epigenetic modifications, and WASp, a potential drug targeting epigenetic pathways to prevent transcription-associated DNA damage in malignant tumors by addressing epigenetic deregulation, such as clearing the R-loop. These approaches have been shown to promote the differentiation of Th cells in T-cell acute lymphoblastic leukemia (T-ALL) patients and inhibit T-ALL progression [125]. However, further research is needed to fully understand the complex interactions between cancer development mechanisms, sex-specific factors, and T cell epigenetics, which may pave the way for novel therapeutic strategies.
3. Sex bias in B cell tumor immunity
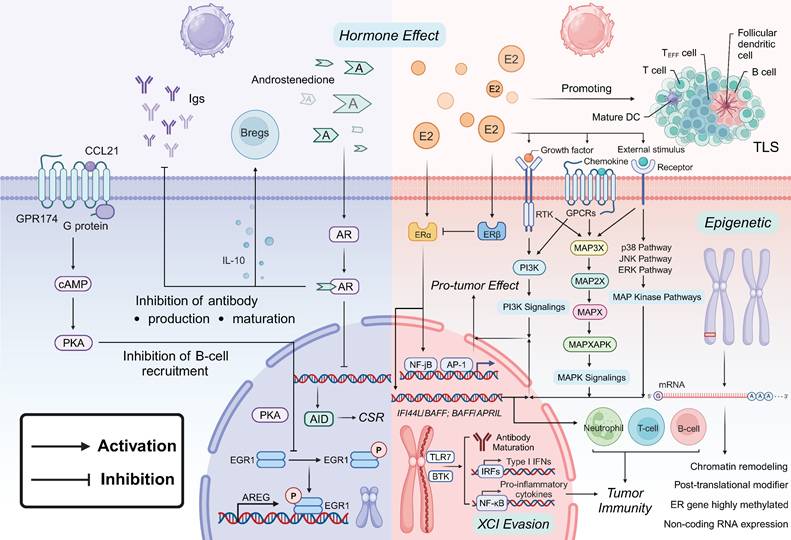
In disease models of SLE in females and males, including the Klinefelter syndrome model, B cell function is modulated by a range of molecular mechanisms, including sex hormones, sex chromosomes, and epigenetic factors [50, 126-128]. These differences indicate that B cell-mediated immune responses in the TME may exhibit significant sex disparities (Figure 2).
Sex Factors in Regulating B Cell Tumor Immunity. Hormone Effects on B Cells: Estrogen interacts with its respective receptors—ERα and ERβ, to promote B cell activation, antibody production, and maturation within the TME. Estrogen enhances growth factor receptor signaling and activates downstream PI3K and MAPK pathways, which contribute to pro-tumor effects, including the promotion of TLS. In males, androgens such as androstenedione act through AR to suppress B cell recruitment, inhibit antibody production, and reduce antibody maturation. The AR pathway negatively regulates B cell function by enhancing IL-10 production in Bregs, which in turn suppresses B cell activation and overall immune responses. Additionally, GPR174 signaling, modulated by the CCL21/GPR174 axis, inhibits B cell aggregation and function within germinal centers, further contributing to the suppression of antibody production and immune responses. XCI Evasion and Epigenetic Pathways: In females, the evasion of XCI results in the overexpression of genes such as TLR7 and BTK, which are crucial for B cell proliferation and activation. The escape of TLR7 from XCI, when combined with estrogen signaling, upregulates the IFI44L/BAFF pathway, thereby promoting IFN-I responses, enhancing immunoglobulin maturation and systemic autoimmune diseases and contributing to heightened inflammatory activation. Sex hormones drive significant epigenetic modifications in B cells, including ER gene methylation, post-translational modifier, non-coding RNA expression and chromatin remodeling. These changes influence gene expression, impacting tumor immunity and potentially leading to developing anti-estrogen resistance in cancer, translational repression of mRNAs, endocrine resistance, and drug resistance.
3.1 Sex bias in TLS
Tumor-infiltrating B lymphocytes (TIL-Bs), comprising both tumor-infiltrating B cells and plasma cells, play a critical and multifaceted role in tumor immunity and are associated with improved clinical outcomes. These B cells contribute to the formation of tertiary lymphoid structures (TLS), which are aggregates of immune cells resembling lymph nodes and serve to establish localized and sustained tumor immune responses [129]. TIL-Bs inhibit tumor progression through multiple mechanisms, including the secretion of immunoglobulins, which promote T cell responses, as well as cross-presenting antigens to T cells to either activate or directly kill them [129-131]. Within TLS, TIL-Bs may function as antigen-presenting or regulatory cells, thereby influencing T cell activity [129]. They are also involved in the formation, maturation, and maintenance of TLS, which has been shown to reduce tumor growth [132]. The presence of high concentrations of B cells, particularly in TLS, is associated with better prognosis and improved responses to ICIs [133, 134], likely due to the TLS-induced infiltration of CTLs into the tumor [135, 136]. Clinical studies indicate that the extent of TIL-B infiltration, particularly in breast cancer, is positively correlated with favorable outcomes. This highlights the potential of inducing tumor immunoreactivity in TIL-Bs through modulation of other immune cell subpopulations as a promising strategy for immunotherapy [137, 138].
Sex differences in B-cell-mediated tumor phenotypes are frequently observed in TMEs characterized by TLS versus B-cell tumors. These differences are believed to be mediated by sex hormone pathways, XCI, gene expression regulation, and epigenetic mechanisms. In patients with papillary thyroid carcinoma (PTC) and NSCLC, a higher area ratio of immune cell aggregates is observed in females, leading to more effective tumor immune surveillance and enhanced anti-tumor immunity [139]. This may be attributed to the presence of more mature and diversified TLS in females, facilitated by sex hormone pathways. In pancreatic adenocarcinoma (PAAD), a significant positive correlation has been observed between the levels of estrogen receptors ESR1 and ESR2 and TLS scores in the TME. Female patients with PAAD show higher TLS scores and a better prognosis, suggesting that estrogens may play a pivotal role in promoting TLS formation and enhancing anti-tumor immunity [140]. These TLS play a crucial role in the regulation of immune cell function and in supporting tumor antigen-driven activity within the TME [139].
3.2 Estrogen regulation of B cell malignancy
In B-cell lymphoma, female patients generally report a higher quality of life, experiencing reduced fatigue, pain, and anxiety/depression compared to their male counterparts [141]. However, in diffuse large B-cell lymphoma (DLBCL), prognosis and survival rates decline significantly after menopause, suggesting that estrogens may have a regulatory effect on B-cell tumors [142]. Estrogen's role in B-cell malignancies likely involves activation of downstream signaling pathways such as MAPK, PI3K, and the MAP kinase family. Estrogen has been shown to increase B-cell activating factor (BAFF) transcript levels in B cells, suggesting that estrogen-induced upregulation of IFI44L/BAFF or BAFF/APRIL expression may activate B cells, T cells, and neutrophils, contributing to immune modulation [143-146]. Chimeric antigen receptor T-cell (CAR-T) therapies targeting B-cell cancers based on BAFF ligands have shown notable success [147]. However, the BAFF/APRIL system can also act as an inhibitor of tumor infiltrating CD8+ T cells, inhibited by regulatory B cells (Bregs), and a pro-oncogenic factor in some tumors, complicating its role in tumor progression [145, 148].
Additionally, ER signaling regulates gene transcription through interactions with transcription factors like AP-1 and NF-jB [149]. ERα-related signaling pathways promote cell proliferation and tumorigenesis, while ERβ typically acts antagonistically [150]. Agonists and estrogen analogues that activate ERβ can inhibit angiogenesis and cancer cell proliferation, suppressing cancer development, except in chronic lymphocytic leukemia (CLL) [151, 152]. ERβ-targeted antagonists, such as tamoxifen, have been shown to significantly induce cell death in DLBCL and reduce the risk of DLBCL in patients with ERα+ breast cancer, highlighting the therapeutic potential of targeting ERβ in B-cell lymphoma treatment [142].
3.3 Androgen regulation of B cell malignancy
Statistical analyses of DLBCL patients indicate that male gender is a negative prognostic factor for overall survival (OS) and progression-free survival (PFS). Men typically exhibit poorer prognosis and higher recurrence rates, likely due to the negative effects induced by androgens [153]. In animal models, such as androgen receptor knockout (ARKO) mice, DHT has been shown ineffective in restoring B cell function. These mechanisms may involve androgen receptor signaling promoting gastric cancer (GC) cell genesis and progression through overexpression of miR-125b and inhibition of apoptosis [154]. Similar mechanisms have been identified in bladder cancer as well [155]. Furthermore, B cells in testicular feminization (Tfm) mice do not respond to androgens in bone marrow-derived cells (BMDCs) [156], indicating that androgen regulation of B cell function occurs via AR and possibly alternative pathways. One such pathway involves the X chromosome-encoded G protein-coupled receptor-174 (GPR-174), which is involved in the CCL21/GPR signaling axis. This signaling pathway inhibits B cell aggregation in the germinal centers of male mice, further emphasizing the complex role of androgens in modulating immune responses and cancer progression [157]. In ER+ breast cancer, androgens inhibit ER-dependent cell proliferation while simultaneously upregulating BCL2 family proteins. This dual action has been associated with a more favorable prognosis in anticancer treatments, suggesting that androgen signaling might modulate the balance between pro-survival and pro-apoptotic factors in certain cancers [158].
3.4 Sex chromosome and epigenetic regulation of B cell malignancy
In nearly 90% of patients with human B-cell leukemia and lymphoma, as well as other significant tumors, high methylation of ER genes and low ER expression, along with noncoding RNA expression, chromatin remodeling, and posttranslational modifications, are observed [159-162]. These factors contribute to translational repression of mRNAs, leading to endocrine resistance and drug resistance. Therapeutic strategies involving histone acetyltransferases (such as MYST3), deacetylases (like HDAC1), and demethylases (e.g., LSD1 and miR-29b) may help restore sensitivity to estrogens and ER expression, offering a potential avenue for overcoming resistance and improving treatment outcomes [162]. In patients with DLBCL undergoing rituximab immunotherapy combined with chemotherapy, PFS significantly improves in women [163], which may be linked to sex differences in adipose tissue distribution [164].
4. Sex bias in NK cell tumor immunity
4.1 Estrogen regulation of NK cells
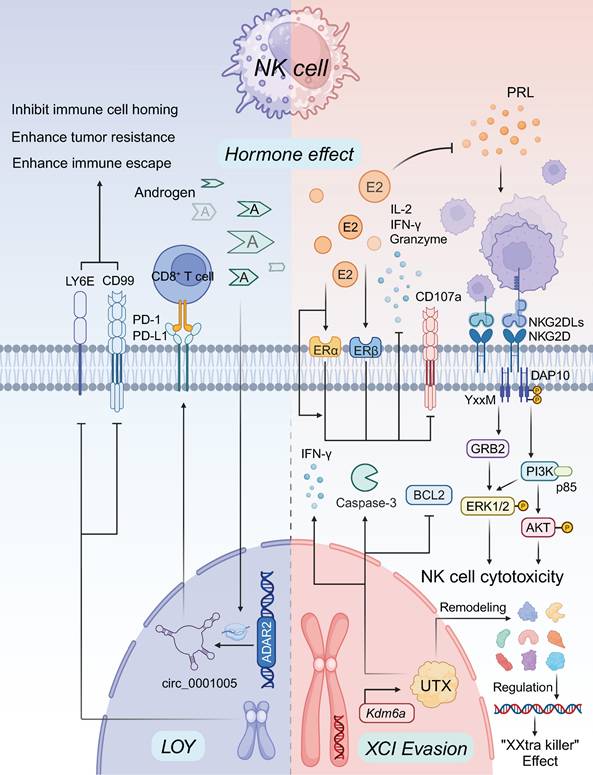
The influence of estrogen on NK cells is complex and multifaceted, impacting various aspects of their function. Estrogen regulates NK cell proliferation, cytotoxicity, secretion of granzyme B, production of cytokines such as IL-2 and IFN-γ, and the expression of surface molecules like CD107a [64, 165-167]. These effects occur through both ER-dependent and ER-independent mechanisms. NK cells express both ERα and ERβ on their surface, with the latter typically recognized as the negative target of estrogen's effects through a non-ERα-mediated pathway [168]. However, recent research suggests that estrogen's impact on NK cell in peripheral blood activity may vary depending on the specific NK cell subtype [169]. For example, estrogen has been shown to enhance the immune response of NKT cells [39]. Estrogen deprivation therapies, such as selective estrogen receptor modulators (SERMs), have demonstrated promise in cancer treatment by stimulating NK cell and CD8+ T cell activation, particularly in peripheral blood from triple-negative breast cancer (TNBC) patients [170]. In contrast, prolactin (PRL) has been found to enhance NK cell-mediated cytotoxicity in cervical cancer through the NKG2D/NKG2DL axis, which may counterbalance the effects of estrogen [171]. While prolactin receptor signaling on NK cells is recognized, its influence is more prominent in conditions like recurrent miscarriages rather than in cancer-related contexts [172] (Figure 3).
Sex Factors in Regulating NK Cell Tumor Immunity. E2 impacts on NK cell functions through its estrogen receptors, ERα and ERβ, including proliferation, cytotoxicity, granzyme B secretion, and the synthesis of IL-2 and IFN-γ. E2 also influences the expression of surface molecules like CD107a through both ER-dependent and non-ER-dependent pathways, depending on NK cell subtypes and context, with estrogen sometimes enhancing the immune response of NKT cells. PRL, has been observed to promote NK cell-mediated cytotoxicity in cervical cancer via the NKG2D/NKG2DL axis, counteracting some of the inhibitory effects of E2 on NK cell function. Androgen signaling is known to suppress NK cell immune responses, potentially by upregulating PD-L1. In models of antiandrogen treatment and AR knockout, downregulation of the ADAR2 gene product circRNA circ_0001005 in NK cells led to reduced PD-L1 expression, which indirectly enhanced NK cell-mediated tumor killing through increased CD8+ T cell activity. The KDM6A gene, evades XCI, leading to elevated UTX levels. UTX plays a crucial role in preventing NK cell quiescence and enhancing effector functions by remodeling chromatin and regulating gene expression. UTX-deficient NK cells show increased expression of the anti-apoptotic factor BCL2 and decreased IFN-γ production, leading to reduced NK effector function. LOY in NK cells is associated with downregulation of key immune homing molecules like CD99 and LY6E, which can impair NK cell-mediated immune surveillance and promote tumor immune escape.
4.2 Androgen regulation of NK cells
In contrast, androgen signaling is known to suppress NK cell immune responses. The blockade of AR has gained attention as a potential strategy for cancer therapy. In a study involving antiandrogen treatment and ARKO models, Qing Liu et al. discovered that the circRNA circ_0001005, encoded by the ADAR2 gene, was downregulated in NK cells, leading to reduced PD-L1 expression. This decrease in PD-L1 facilitated NK cell-mediated tumor elimination by enhancing CD8+ T cell activity [173, 174]. The inhibitory effect of androgens on NK cell cytotoxicity may involve upregulation of PD-L1 or suppression of IL-12A expression in tumor cells. Evidence suggests that androgen receptor antagonists improve tumor control in mouse models of bladder cancer [175-177]. However, further research is needed to fully understand the direct impact of androgens on NK cell function.
4.3 Sex chromosome and epigenetic regulation of NK cells
Sex differences in NK cell activity are influenced not only by sex hormones but also by sex chromosomes and epigenetic factors. Studies have shown that female NK cells persistently secrete higher levels of IFNγ, perforin, and granzyme B compared to male NK cells in gonadectomized mice, suggesting that X-linked epigenetic regulators contribute to NK cell sex bias [178]. Recent research has shifted focus from gonadotropins to the X-linked epigenetic regulator, ubiquitously transcribed tetratricopeptide repeat on chromosome X (UTX), which is encoded by the KDM6A gene [179]. The KDM6A gene evades XCI, leading to elevated UTX protein levels. UTX regulates gene expression by remodeling chromatin, thus preventing NK cell quiescence and enhancing their effector functions, effectively making female NK cells "XXtra killer cells" [180, 181]. In contrast, UTX-deficient NK cells show increased expression of the anti-apoptotic factor BCL2 and decreased IFNγ production, resulting in a higher frequency of NK cells but impaired effector function [50]. Additionally, both male and female UtxNKD cells exhibit altered caspase-3 activity and increased BCL2 expression, suggesting that UTX plays a critical role in NK cell homeostasis by promoting apoptosis [179]. LOY has been linked to increased tumor incidence, aggression, and metastasis, potentially due to the downregulation of CD99 and LY6E in NK cells [118, 182]. This downregulation may impair immune cell homing, thereby enhancing tumor resistance and facilitating immune escape [183]. Additionally, epigenetic factors, including DNA methylation, histone modifications, transcription factors, and miRNA regulation, play significant roles in modulating NK cell-mediated cytotoxicity [184]. However, the precise functions of these factors, particularly in relation to sex-based differences, remain to be fully elucidated and warrant further investigation.
5. Sex bias in dendritic cell tumor immunity
While research on sex bias in dendritic cells (DCs) within the TME is limited, sex-specific disparities in DCs are still largely influenced by sex hormones pathways and other related factors, which may provide us innovative insights in DCs cancer immunology (Figure 4).
Sex Factors in Regulating DCs Tumor Immunity. Higher concentrations of P4 regulate DC phenotypes through interactions with PR signaling pathways, associated with the upregulation of TLR7, Unc93b1, and TLR9 in DCs, which enhances their activation and phagocytic abilities. P4 also correlates with increased levels of IFN-α+ pDCs in peripheral blood, which can inhibit TNF and IL-1β secretion by conventional DCs and downregulate their ability to activate T cells. E2 exerts similar effects by increasing the proportion of IFN-α+ DCs through upregulation of Unc93b1 and IRF5 expression via ERα dependent pathways. E2/ERα signaling enhances DC activation by upregulating MHC class II molecules and co-stimulatory molecules, thereby boosting their APCs efficacy. This signaling also increases environmental GM-CSF and IRF levels, further augmenting DC-mediated immune responses. The enhanced TLR7 and IFN signaling in female DCs, due to XCI evasion, contributes to the heightened production of IFN-α, particularly in pDCs. AR-mediated signaling exerts inhibitory effects on DC function, particularly in T cell activation and pDC responses. Androgens suppress the surface expression of MHC-II and co-stimulatory molecules (e.g., CD80, CD83, CD86, CD40, OX40L) on DCs, leading to reduced capacity to stimulate T cells. High androgen levels in male infants and adolescents are associated with lower IFN-α production by pDCs, and testosterone administration in transgender males also diminishes pDC production of IFN-I, further emphasizing the suppressive role of androgens on DC-mediated immunity. XCI evasion results in the overexpression of X-linked genes such as TLR7, CYBB, and IL13RA1 in DCs, enhancing DC activation, phagocytosis, and IFN-α production, contributing to a more robust immune response compared to males.
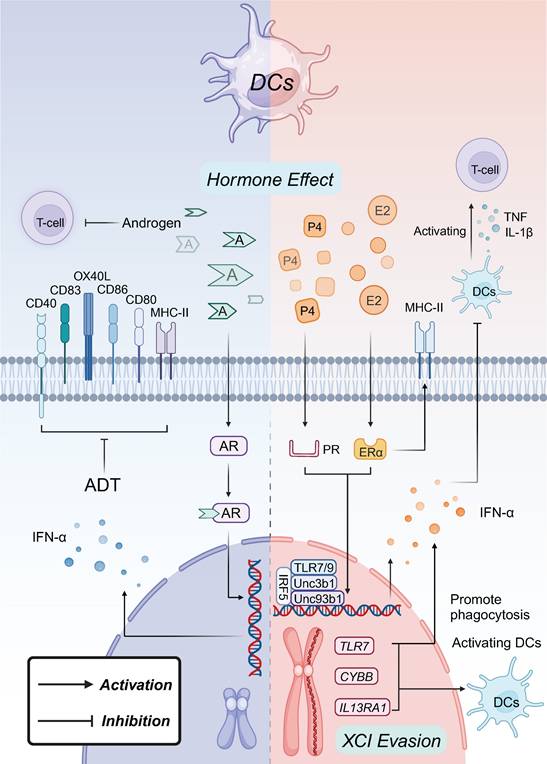
5.1 Female sex marks regulation of DCs
P4, which is present at higher levels in females, plays a key role in regulating DCs phenotype through interactions with both the PR and the glucocorticoid receptor (GR) signaling pathways. Studies have shown a correlation between elevated serum P4 levels and increased peripheral blood levels of IFN-α+ plasmacytoid dendritic cells (pDCs) [185]. These pDCs inhibit the secretion of tumor necrosis factor (TNF) and IL-1β by DCs and downregulate their ability to activate T cells. This effect is likely due to the P4/PR and P4/GR signaling axes, which upregulate TLR7, Unc93b1, and TLR9 in female-derived DCs [186, 187].
Estrogen may exert similar effects on DCs by increasing the proportion of IFN-α+ DCs activated through TLR7 or TLR9 stimulation. This process involves the upregulation of Unc93b1 and IRF5 expression via estrogen receptor 1 (ESR1)-dependent pathways [39]. Additionally, the ability of DCs to regulate the activation of other immune cells, as opposed to responding to immune cell activation, exhibits sex differences, primarily mediated by E2 and estrogen ERα signaling. E2/ERα signaling upregulates major histocompatibility complex (MHC) class II molecules and co-stimulatory molecules on DCs, enhancing their activation. This signaling also increases environmental GM-CSF and IRF levels, further boosting the antigen-presenting cells (APCs) efficacy [39].
In about 1% of severely ill male COVID-19 patients, defects in TLR7 have been identified, impairing the ability of pDCs to secrete IFN-I and provide protective immunity. This defect is linked to a variant of the TLR7 gene located on the X chromosome [188]. Additionally, increased TLR7 expression and heightened IFN signaling, influenced by X chromosome dosage, support the hypothesis that TLR7 escapes XCI, with estrogen further enhancing this effect [189, 190]. As a result, female pDCs produce higher levels of IFN-α in response to TLR7 signaling compared to males, a difference that contributes to the pathogenesis of SLE and HIV-1 infection [191]. Moreover, IL13RA1 and CYBB have also been shown to evade XCI in DCs, leading to enhanced DCs activation and phagocytosis [192]. While the specific gender-differentiated effects within the TME remain under investigation, DCs appear to activate T-cell-dominated tumor immune responses more effectively in females, leading to improved tumor healing and prolonged survival.
5.2 Male sex marks regulation of DCs
In males, AR-mediated inhibitory effects on DCs primarily impact T cell maturation, differentiation, and pDCs function. Male infants, during early responses to viral infections when androgen levels are high, produce significantly lower levels of IFN-α from pDCs compared to females [193]. Similarly, in adolescents (12-16 years of age) following mRNA vaccination, androgens delay pDCs activation and reduce IFN-α production, with this effect negatively correlating with free testosterone levels [194]. Testosterone administration in transgender males also results in decreased IFN-I production by pDCs [195]. In androgen-deprived male mice, surface expression of MHC-II and co-stimulatory molecules like CD80, CD83, CD86, CD40, and OX40L is downregulated on DCs in prostate cancer [196, 197]. Direct exposure to dihydrotestosterone impairs activated T cell function in DCs [198]. Furthermore, young male rats exhibit a higher proportion of immature DCs and a reduced ability to stimulate CD4+ T cell polarization compared to females [199]. This observation is supported by studies in rhesus monkeys with leukemia, which show a lower number of myeloid DCs in males than in females [200]. The absence of sex-specific differences in AR-deficient DCs suggests that androgen effects on DCs are AR-dependent [200].
Furthermore, sex differences significantly influence the phenotype of DCs across various diseases, with both estrogen and androgen receptors present in cancer tissues [201]. However, despite these distinctions, significant sex differences have not been observed in the effectiveness of cancer therapies, such as DCs vaccine therapy [202]. The application of sex-based strategies for tailored cancer treatment is still challenging at the level of DCs function and response.
6. Sex bias in Neutrophil tumor immunity
Neutrophils within tumor show notable sex-based heterogeneity, driven primarily by sex hormones, sex chromosomes and epigenetic factors. This variability impacts several key neutrophil functions, including activation, apoptosis, proliferative infiltration, surface checkpoint expression, and the formation of neutrophil extracellular traps (NETs) (Figure 5).
Sex Factors in Regulating Neutrophils Tumor Immunity. E2 plays a pivotal role in neutrophil function by promoting differentiation, inhibiting apoptosis, and enhancing NET and NETosis. In renal cell carcinoma, estrogen facilitates neutrophil infiltration into the TME via ER signaling and the VEGFα/HIF2α pathway, contributing to tumor progression. Additionally, estrogen inhibits neutrophil degranulation, which can suppress tumor immunoreactivity through the PD-1/PD-L1 pathway and affect oxygen-dependent intracellular killing mechanisms. Estrogen also depends neutrophils in female exhibiting a greater number of mallet bodies. HCG enhances NET formation, which can either promote tumor cell death or contribute to metastasis, depending on the context, highlighting its complex role in tumor biology. P4 acts as an antagonist to NETosis by inhibiting the transfer of NE from the cytoplasm to the nucleus, reducing NET formation and impacting neutrophil-mediated tumor immunity. Testosterone promotes the anti-tumor effect of neutrophil by enhancing its cytotoxic and proliferation, while loss of testosterone inhibits endogenous analgesia and increases tumor load. ADT impairs neutrophil function by inhibiting cytotoxic and proliferation through the increased expression of TβRI, thereby reducing the tumor immunoreactivity of neutrophils and PMN. Epigenetic modifications involving the X chromosome significantly influence neutrophil function. TLR7 and TLR9 genes, which evade XCI, are upregulated in neutrophils, enhancing their recruitment and activation through the TLR7-MyD88-DC pathway. The CXCL16 axis and TLR7/8 agonists further promote neutrophil recruitment, potentially leading to antitumor effects.
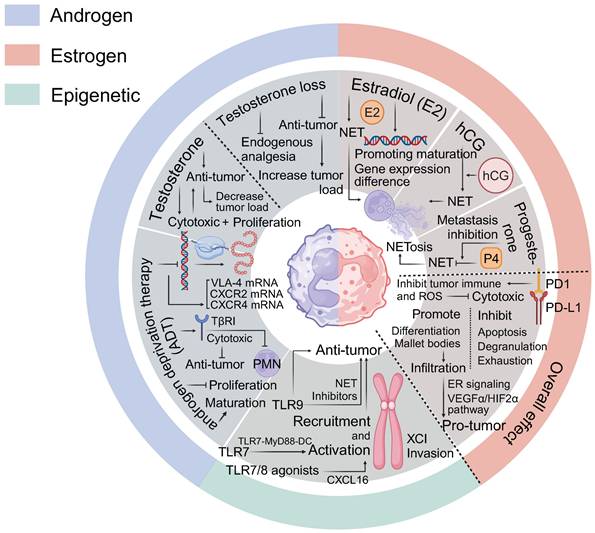
6.1 Estrogen regulation of Neutrophils
In hepatocellular carcinoma (HCC), neutrophils contribute to tumor progression through various mechanisms [203]. For example, neutrophils in this context express PD-L1, which impairs tumor immune responses. Single-cell RNA sequencing(scRNA-seq) studies have demonstrated differential gene expression in E2-stimulated neutrophils, shedding light on how estrogen influences infiltrating macrophages and monocytes [204]. Furthermore, mRNA sequencing has revealed that male neutrophils are generally more immature and metabolically active than their female counterparts [205]. Estrogen plays a key role in promoting neutrophil differentiation and inhibiting apoptosis, leading to observed sex differences in neutrophil numbers and functionality [206, 207]. Additionally, estrogen has been shown to inhibit neutrophil degranulation, which can suppress tumor immunoreactivity through the PD-1/PD-L1 pathway and impact oxygen-dependent intracellular killing mechanisms [208].
Both E2 and hCG have been shown to enhance NETs formation and NETosis. In contrast, P4 acts as an antagonist, inhibiting the efficient transfer of neutrophil elastase (NE) from the cytoplasm to the nucleus, thereby impeding NET formation [209]. In the context of neutrophil exhaustion and the inhibition of NETosis, estrogen has been implicated in promoting aneurysm rupture. Conversely, estrogen-deficient mice demonstrate a decreased likelihood of aneurysm rupture [210]. Estrogen also contributes to the sex-differentiated appearance of neutrophils, with females displaying a greater number of mallet bodies [211]. Furthermore, estrogen facilitates neutrophil infiltration into the TME by modulating cellular communication through the ER pathway. This process plays a role in promoting renal cell carcinoma progression, with infiltrating neutrophils contributing to tumor advancement, particularly via ER signaling and the VEGFα/HIF2α pathway [212, 213].
6.2 Androgen regulation of Neutrophils
Androgens have been shown to negatively impact neutrophil function in the TME, but this perspective may not capture the full scope of their effects. GC illustrates a notable sex disparity, with incidence and mortality rates in males approximately twice as high as in females. Statistical analyses have revealed that tumor-associated neutrophil (TAN) density varies by GC type, with diffuse GC exhibiting the least TAN infiltration. Importantly, male patients demonstrate greater variability in TAN density within tumors compared to females, suggesting that TAN density may serve as a sex-specific prognostic indicator for GC [214, 215].
In melanoma models, male mice subjected to gonadal castration showed increased tumor burden, while testosterone treatment in female mice reduced tumor sizes. Furthermore, neutrophils from castrated male mice exhibited lower percentages and expressed reduced CXCR2 mRNA levels, alongside higher CXCR4 and VLA-4 mRNA levels, impairing neutrophil activation and immune responses [216]. These findings underscore the influence of sex hormones on neutrophil numbers, gene expression, and cytotoxic functions [216]. Additionally, in renal infections and inflammation, androgen exposure has been associated with impaired neutrophil maturation and function [217]. Similarly, testosterone antagonists used in oral cancer models have been shown to mimic opioid effects, impacting neutrophil-mediated analgesia [218].
The observed reduction in neutrophil numbers and altered function following castration of male mice, compared to the myeloid-derived suppressor cell-like phenotype in prostate cancer patients undergoing ADT, emphasizes the complex role of androgens in modulating immune responses [216]. These findings suggest that androgen-mediated changes in neutrophil function may contribute to tumor metastasis and progression [216]. Further studies on ADT have revealed abnormalities in neutrophils from both ADT-treated patients and castrated male mice. ADT appears to impair neutrophil-mediated antitumor activity by inhibiting cytotoxic functions, likely through the increased expression of transforming growth factor β receptor I (TβRI) [219]. This inhibition suppresses the tumor immunoreactivity of peripheral blood polymorphonuclear neutrophils (PMNs), potentially reducing their effectiveness in combating tumor growth [219].
6.3 Sex chromosome and epigenetic regulation of Neutrophils
The observed phenotypic differences in neutrophils can be attributed to epigenetic modifications, particularly those involving the X chromosome. Studies have shown that TLR7 genes, by evading XCI, can upregulate their own expression, thereby enhancing neutrophil recruitment and activation through the TLR7-MyD88-DCs signaling pathway. Similarly, the CXCL16 axis and TLR7/8 agonists are effective in recruiting large numbers of neutrophils, potentially contributing to antitumor effects [220, 221]. TLR9 agonists have also demonstrated substantial anticancer efficacy across various types of cancer, with TLR9 expression in neutrophils emerging as a crucial prognostic marker [222, 223]. Notably, TLR9, which also evades XCI, interacts with NETs to inhibit Merlin phosphorylation, leading to resistance to ferroptosis in TNBC cells. Consequently, targeting NETs appears to be a promising therapeutic strategy for TNBC [224].
7. Sex bias in Macrophage tumor immunity
Androgens, estrogens, Sex chromosomes and epigenetic factors have a profound impact on macrophage function in tumor, influencing both cancer progression and immune responses through various mechanisms (Figure 6).
Sex Factors in Regulating Macrophages Tumor Immunity. (A) In melanoma, downregulation of AR signaling enhances macrophage infiltration, inhibits tumor progression, and may provide a basis for novel immunotherapeutic strategies. In prostate cancer, androgens such as testosterone and dihydrotestosterone activate STAT3 and upregulate genes like TREM-1, CCL2, CCL7, CCL13, and CXCL8, promoting metastasis. Additionally, in thyroid cancer, testosterone influences oncogene expression (e.g., CD52, Sh2d1b1, Fcgr3, Itgam, Glipr1, Sfrp1) and inhibits immune cell infiltration while SupraT can induce immune responses through ferritin and nuclear autophagy and thus through nucleic acid sensing-NFκB signaling in prostate cancer. AR signaling also promotes TRAIL gene transcription, enhancing macrophage cytotoxicity against prostate cancer cells. (B) In NSCLC, ERα activation drives the CCL2/CCR2 axis, promoting macrophage infiltration and cancer cell invasion, while upregulating CXCL12/CXCR4 signaling. Estrogen inhibits DNMT1, leading to decreased p53 expression and inducing M2 macrophage polarization. In endometrial cancer, IL-17A-mediated epigenetic mechanisms induce ERα expression in macrophages, which sensitizes cancer cells to estrogen and promotes tumor progression. Estrogen also upregulates CD47, inhibiting macrophage phagocytosis in ER+ tumor cells; blocking CD47 with anti-estrogens enhances macrophage phagocytosis. In ER+ breast cancer, estrogen stimulates macrophages through CCL2 and CCL5, contributing to tumor progression. (C) Female bone marrow-derived macrophages with deletion of the X-linked gene Ddx3x exhibit impaired innate immune responses, including reduced cytokine production (IL-1, IL-6, IL-12, TNF) and a diminished ability to restrict bacterial proliferation. The Kdm6a and p53 gene, and miRNA-223-3p, which promotes M2 macrophage polarization, contribute to bladder carcinogenesis. The LOY disrupts the Y-linked gene Uty, promoting pro-fibrotic macrophage phenotypes.
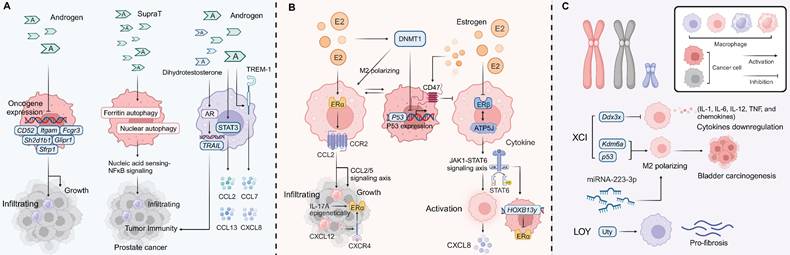
7.1 Androgen regulation of macrophages
AR signaling plays a key role in regulating macrophage infiltration, cytotoxicity, and immune responses. In melanoma, downregulation of AR signaling has been shown to increase macrophage infiltration, inhibit tumor progression, and offer potential avenues for immunotherapy [225]. In contrast, AR signaling can enhance macrophage-driven promotion of breast cancer progression [226]. In prostate cancer, androgen stimulation mediates metastasis through STAT3 activation and the upregulation of inflammatory cytokines such as TREM-1, CCL2, CCL7, CCL13, and CXCL8 [227, 228]. Additionally, in thyroid cancer, ThrbPV/PV mice, testosterone influences oncogene expression, including CD52, Sh2d1b1, Fcgr3, Itgam, Glipr1, and Sfrp1, and inhibits macrophage and other immune cell infiltration [229]. In colorectal and prostate cancer, males are more biased to show more anti-tumor M1 polarization while more M2 phenotype is demonstrated in females [24, 228]. Supraphysiologic levels of testosterone (SupraT) can induce immune responses by activating ferritin and nuclear autophagy, subsequently triggering nucleic acid sensing-NFκB signaling in prostate cancer [230]. Moreover, dihydrotestosterone promotes the transcription of TRAIL genes via AR signaling, enhancing macrophage cytotoxicity against prostate cancer cells [231]. A similar effect and sex bias have also been observed in immune checkpoint blockade (ICB) therapy for melanoma, where male patients derive greater benefit from ICB treatment compared to females, primarily influenced by the functionality of intratumoral macrophages [232].
7.2 Estrogen regulation of macrophages
Estrogen regulates macrophage activity and tumor progression through interactions with a wide range of cytokines and signaling pathways. Estrogen upregulates CD47 in ER+ tumor cells, inhibiting macrophage-mediated phagocytosis [233, 234]. Inhibition of CD47 with anti-estrogens has been shown to enhance macrophage phagocytosis, suggesting a potential therapeutic strategy [234]. Additionally, estrogen promotes tumorigenesis in normal breast tissue by increasing the number of CD206+ macrophages, while simultaneously reducing the presence of dendritic cells and T cells. Conversely, oophorectomy, which reduces estrogen levels, leads to an increase in M1 macrophages and has been associated with slowed progression in ovarian cancer [235, 236].
ER signaling enhances tumor immunity by reducing immune checkpoint resistance, with tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs) playing a central role in this process. In endometrial cancer, uterine-infiltrating CD68+CD163+ macrophages induce ERα expression through IL-17A-mediated epigenetic mechanisms, sensitizing cancer cells to E2 and accelerating tumor progression [237]. In breast cancer, estrogen stimulates macrophage activity via CCL2 and CCL5, while in endometrial cancer, macrophage-induced ERα expression enhances tumor progression [238]. In HCC, estrogen represses tumor growth by inhibiting the interaction between ERβ and ATP5J, blocking the JAK1-STAT6 axis, and inhibiting TAM alternative activation, which in turn secretes CXCL8. This leads to the loss of ERα in endometrial carcinoma (EC) through HOXB13y expression, contributing to a poor prognosis [239, 240]. In NSCLC, ERα activation triggers the CCL2/CCR2 axis, promoting macrophage infiltration and facilitating cancer cell invasion, while also upregulating the CXCL12/CXCR4 signaling pathway [241]. Furthermore, a series of reciprocal positive feedback loops between macrophages and cancer cells have been observed in both ER+ breast cancer and TNBC patients [242-244].
Estrogen-induced inhibition of DNMT1 results in decreased p53 expression, which in turn promotes M2 macrophage polarization, contributing to a poor prognosis in lung cancer [245]. Estrogen/ERα-induced infiltration of TAMs and M2 polarization have also been shown to promote cancer progression [246], which promotes CD8+ T cell dysfunction and exhaustion and ICB resistance [232]. Additionally, a study highlighted the potential therapeutic utility of tamoxifen in suppressing brain metastasis of hormone receptor-deficient breast cancer by blocking M2 polarization of microglia and enhancing their anti-tumor phagocytic activity [247]. Exogenous E2 treatment reduces the pro-inflammation phenotype in macrophage cells and accelerates resolution of inflammation through IL-10 activation [64]. Interestingly, 2-methoxyestradiol (2ME2), a metabolite of 17β-estradiol, has been shown to suppress M2 polarization and the pro-tumoral functions of macrophages in breast cancer by inhibiting STAT3. This suggests that further investigation is still needed to explore the potential estrogen-related pathways in macrophage polarization [248].
7.3 Sex chromosome and epigenetic regulation of macrophages
In addition to sex steroid hormone-mediated differences in macrophage immunity, sex chromosome and epigenetic factors also play a significant role in the observed sex variability in macrophage immune responses. For example, female bone marrow-derived macrophages with a deletion of the X-linked gene Ddx3x exhibit impaired ability to restrict the proliferation of Listeria monocytogenes and show a marked reduction in the expression of key cytokines, such as IL-1, IL-6, IL-12, TNF, and various chemokines [50]. This deficiency in Ddx3x disrupts their functional response in innate immunity. Moreover, the Kdm6a gene, which has oncogenic properties when overexpressed in NK cells, has been found to be deficient in macrophages. This deficiency promotes M2 macrophage polarization and contributes to bladder carcinogenesis, particularly in the context of p53 deficiency [249]. On the other hand, bladder cancer cells can be effectively inhibited by miRNA-223-3p, an X-chromosome-linked miRNA that regulates macrophage polarization. miRNA-223-3p reduces M1 levels, thereby diminishing the inflammatory response. This results in a relative disadvantage for females in terms of innate immune responses, due to the escape mechanisms of XCI that allow for the upregulation of immune-modulatory genes like miRNA-223-3p [250]. Additionally, the loss of Y chromosome chimerism, which is often associated with changes in the X chromosome, leads to the disruption of the Y-linked gene Uty. This disruption has been found to promote pro-fibrotic macrophage phenotypes, which in turn contribute to the development and progression of sarcoma [251, 252].
8. Sex bias in other immune cells and pathways
Despite limited research on sex disparities in immune cells like MDSCs, innate lymphoid cells (ILCs), mast cells, eosinophils, and basophils within tumor immunology, sex-dependent biases are evident.
MDSCs associated with chronic inflammation include both mononuclear MDSCs (mMDSCs) and granulocytic MDSCs (gMDSCs). These cells primarily inhibit CTLs and NK cells within the TME, thereby promoting tumor angiogenesis, invasion, and metastasis [253, 254]. In female patients, a higher level of peripheral gMDSCs is linked to poor prognosis, whereas in males, tumor-infiltrating mMDSCs are more prominent [255]. E2 facilitates the accumulation of MDSCs in the bloodstream by stimulating the secretion of TNF-α in vivo [254]. Additionally, E2-treated MDSCs, particularly mMDSCs, interact with T cells in melanoma, leading to T cell exhaustion and reduced cytotoxic function [232]. In contrast, androgens downregulate MDSCs, thereby diminishing myeloid cell-mediated immune suppression and inhibiting tumor progression [256]. However, following ADT, MDSC levels increase, which poses a persistent challenge and contributes to the progression of many patients to castration-resistant prostate cancer (CRPC) [256, 257].
Estrogen enhances ILCs tissue infiltration, infection resistance, and cytokine secretion, while androgens inhibit these functions via ER/AR pathways [39, 258-264]. In tissue immunity, the androgen-ILC2-DCs axis negatively regulates sex differences in skin immunity, with androgens suppressing ILC2s [265]. Estrogen and progesterone pathways in mast cells promote IgE-induced degranulation and leukotriene production, but the androgen effect varies depending on cell sources and subtypes [266]. Eosinophils are influenced by estrogen binding to membrane ERα, activating GPR-1, which triggers immune responses such as proliferation and anti-tumor effects [267-270]. In studies on pancreatic cancer development, eosinophils and ILCs are also crucial targets of sex-based regulation in the cancer-microbiome interactions [271].
Further research is required to identify the sex-dependent epigenetic and chromosomal factors that contribute to the dimorphism of MDSCs and the specific role and mechanisms of sex factors in the sex bias of ILCs, eosinophils, and basophils remains unclear, warranting further investigation. Beyond the estrogen and androgen signaling pathways previously discussed, other sex-differentiated molecules, such as FSH, LH, and CRH, also influence immune responses (Table 1). However, the impact of most molecules on immunity in tumor immunity still needs to be explored.
Other sex biased molecules regulation of immunity.
| Molecules | Dominant sex | Regulation of female/male immunity | Refs |
|---|---|---|---|
| Activin/Inhibin | Female | Impairing tumor immunity by affecting CD8+ T cells. | [337, 338] |
| Bile Acids | Male | Upregulating FOXP3+ Tregs and interfering CRC prognosis. | [339] |
| Catecholamines | Male | Activating T cells, inflammation and autoimmunity. | [340] |
| Collagen | Female | Impairing T cells cytotoxicity, stimulating its migration and M2 polarization. | [341] |
| DHEA | Male | Stimulating Th1 response and Tregs, impairing Th17 response. | [342] |
| FSH | Female | FSH downregulation impairs anti-tumor immunity in males. | [343] |
| hCG | Female | Stimulating lymphocyte secretion of IL-10 while downregulating TNF. | [344] |
| NP | Female | Inhibiting inflammasome activation in immune cells. | [345, 346] |
| OT | Female | Activating T cells while inhibiting the infiltration of neutrophils and macrophages, and suppressing the expression of pro-inflammatory cytokines such as TNF-α and IL-1β. | [347] |
| PRL | Female | Promoting T cells, B cells, NK cells, DCs, etc. activation and response. | [348] |
| VP | Male | Stimulating neutrophils and monocytes, thereby promoting inflammation. | [349, 350] |
CD: cluster of differentiation; CRC: colorectal cancer; FOXP3: forkhead box P3; M2: M2 macrophages; DHEA: dehydroepiandrosterone; Th: helper T cell; FSH: follicle stimulating hormone; hCG: human chorionic gonadotropin; TNF: tumor necrosis factor; NP: natriuretic peptides; OT: oxytocin; IL: interleukin; PRL: prolactin; NK cells: natural killer cells; DCs: dendritic cells; VP: vasopressin.
9. Discussion
This review explores the roles of sex chromosomes, sex hormone levels, and sex hormone receptor expression in modulating immune cell behavior, which varies significantly between sexes and influences treatment responses and adverse effects. In general, females exhibit stronger innate and adaptive immune responses than males [272], which may partially explain the higher mortality rates and shorter survival times observed in male patients [2]. However, males often show more favorable responses to immune checkpoint blockade therapy, while females tend to experience more severe adverse events [12]. Additionally, females typically present with more aggressive, advanced, and refractory disease courses, characterized by higher recurrence rates compared to males. This disparity may be linked to the stimulatory effects of sex hormones on cell proliferation, drug resistance, and the inhibition of apoptosis in cancer cells [77].
Significant sex differences also influence the response to radiotherapy, chemotherapy, and emerging targeted therapies and immunotherapies [273, 274]. Gender-related oxidative stress, regulated by sex hormones, impacts redox state proteins and mitochondrial function, potentially altering the efficacy and toxicity of cancer treatments. This mechanism may contribute to the observed greater resistance to cancer in young females compared to males [275]. Moreover, chemotherapy-induced cardiotoxicity is more prevalent in females, while tyrosine kinase inhibitors are more likely to cause vascular embolism in female patients, potentially due to reduced nitric oxide (NO) synthesis and endothelial dysfunction [276]. Additionally, radiotherapy-induced tissue hypomethylation is more pronounced in male patients [275]. These findings highlight the importance of considering sex as a critical factor when evaluating the risks and benefits of cancer therapies.
Sex steroid levels fluctuate significantly across different life stages, including puberty, menopause, and the reproductive phase, leading to considerable variations in hormone concentrations and signaling through sex steroid receptors [64]. Menstrual and reproductive factors significantly influence cancer risk. Nulliparity, or never having given birth, is a well-established risk factor for several cancers, including luminal breast cancer, ovarian cancer, and endometrial cancer [277-280]. However, the relationship between parity and cancer risk is more complex, especially in Black women, where high parity has been linked to a higher risk of triple-negative breast cancer (TNBC) [281]. While the disparity between nulliparity and ever-parity was traditionally thought to stem from differences in ovulatory cycles, recent studies suggest that parous women may have an increased frequency of mutations in breast stromal cells [282, 283]. The timing of menopause also plays a crucial role in cancer risk. Later menopause is associated with a higher incidence of hormone-related cancers, such as breast, endometrial, ovarian, and colon cancers, while early menopause has been linked to an increased risk of lung cancer [284-286]. For pregnant patients with breast cancer, the 5-year disease-free survival (DFS) rate increases from 65% to 71% if they are not pregnant, and the 5-year OS rate rises from 78% to 81% [287]. Similarly, pregnant women with melanoma tend to have lower survival rates compared to their non-pregnant counterparts [288]. Beyond gynecological cancers, the incidence of lung and colon cancers also rises in perimenopausal female, with poorer prognosis compared to postmenopausal group [289, 290]. In serous ovarian cancer ascites, the premenopausal group shows an increased proportion of T cells, while tumor immunity in the postmenopausal group is more associated with the IL-17 pathway [291]. These findings highlight the alteration of immune cell functions in response to varying estrogens over female life course [64].
Hormone replacement therapy (HRT) is another significant factor. Although HRT increases the risk of breast and ovarian cancers, it has been shown to reduce the risk of esophageal, colon, and gastric cancers [292-296]. Notably, HRT regimens containing both estrogen and progesterone are linked to a higher risk of breast cancer compared to estrogen-only therapies. Additionally, the cancer risk associated with HRT is more pronounced in estrogen receptor-positive tumors than in estrogen receptor-negative ones. Importantly, the increased risk of breast cancer diminishes within two years after discontinuing HRT [297].
Sex chromosomes play a crucial role in regulating tumor immune responses. The X chromosome contains approximately 50 genes involved in immune functions, including those responsible for immune cell identification (FOXP3), cellular activation and intracellular signaling (CD40LG, TLR7, IRAK1, IL13RA1/2, NEMO, TASL, IL9R), immune cell differentiation and proliferation (IL2RG, BTK), and cellular metabolism (OGT) [272]. Furthermore, autosomal genes regulating immune pathways can also be influenced by X chromosome inactivation (e.g., EIF1AX and KDM6A) [298]. The long non-coding RNA X-inactivation-specific transcript (XIST), which is regulated by the oncogene P53, has complex effects on cancer, with both protective and promotive roles [299, 300]. Increased cancer risk in females is partly attributed to the loss or dysfunction of tumor suppressor genes that escape XCI, such as ATRX, CNKSR2, DDX3X, KDM5C, KDM6A, MAGEC3, and other X-inactivating tumor suppressors (EXITS) [20].
Furthermore, developmental XCI results in X-chimerism, enabling the preferential expression of specific mosaic subpopulations of the X chromosome in female cells. This process may enhance the immune system's ability to defend against cancer [301, 302]. In contrast, males exhibit an increased risk of cancer due to the LOY and the extreme downregulation of Y-linked genes (EDY), both of which serve as valuable biomarkers for cancer prediction [303]. Incomplete inactivation of the X and Y chromosomes, along with the overexpression of sex chromosome-linked genes in various immune cells—such as T cells, DCs, and NK cells—can contribute to cellular exhaustion and diminished tumor immunoreactivity [21].
Sex hormones and their receptors play a critical role in orchestrating molecular and cellular processes that influence cancer risk and progression. For example, estrogen signaling has been shown to reduce circulating IL-6 levels and inhibit the secretion of inflammatory cytokines by macrophages and neutrophils, thereby lowering the risk of lung and liver cancer in females [304, 305]. In the adaptive immune system, estrogen can upregulate several genes, such as IFN-γ, IFI6, CX3CL1, CX3CL2, IL-1, IL-5, and IL-16 in T cells, which are associated with enhanced inflammatory and cytotoxic T-cell responses [306]. Estrogen also directly regulates other immune cell types, including B cells, dendritic cells, and pDCs, all of which are promising targets for immunotherapy [307, 308]. However, estrogen's role in cancer progression can be dual. It can promote cancer by increasing intracellular PD-1 expression and decreasing p53 expression [309, 310].
Regarding epigenetic effects, androgens have been observed to reduce DNA methylation in embryonic neural stem cells and liver cells in males, creating epigenetic patterns similar to those seen in cancer cells [311, 312]. Additionally, both estrogen and androgens can contribute to tumorigenesis through the mTOR signaling pathway [313]. Sex hormones also influence cancer progression by promoting endothelial cell proliferation and migration. This occurs through the regulation of angiogenic genes, the stimulation of endothelial nitric oxide synthase (eNOS) production, and the activation of mesenchymal stromal cells to secrete factors like VEGF. These effects are mediated by ERs, PRs, and ARs [79, 314-316].
Numerous drugs targeting sex hormone pathways are approved or in development for cancer treatment, including immunotherapies, chemotherapies, and targeted therapies. These include AR antagonists (biclautamide, nilutamide, darolutamide, apalutamide and enzalutamide), LHRH/GnRH agonists (leuprolide, goserelin and buserelin), LHRH/GnRH antagonists (degarelix, relugolix and abarelix), selective ER modulators and downregulators (tamoxifen, raloxifene and fulvestrant), GPER agonists (LNS8801d), aromatase inhibitors (exemestane, letrozole and anastrozole) [12, 317]. Several clinical trials exploring sex hormone-based therapies in non-reproductive organ-derived cancers have been conducted, with the potential mechanisms of modulating anti-cancer immune responses still under investigation (Table 2).
Registered clinical trials of sex hormone-based therapy conducted in non-reproductive organ derived cancers.
| NCT identifier | Trial titles | Phase | Population description | Status | Cancer type | Publication |
|---|---|---|---|---|---|---|
| NCT00002595 | Toremifene in Treating Patients with Desmoid Tumors | Phase II | n=72 (Estimated); Drug (Target): Toremifene (SERM); Procedure: conventional surgery | Completed | Desmoid Tumor | NA |
| NCT02353429 | Toremifene in Desmoid Tumor: Prospective Clinical Trial and Identification of Potential Molecular Targets | Phase II | n=25 (Estimated); Drug (Target): Toremifene (SERM) | Unknown status | NA | |
| NCT00068419 | Sulindac and Tamoxifen in Treating Patients with Desmoid Tumor | Phase II | n=70; Drug (Target): Tamoxifen citrate (SERM)/Sulindac (COX-1/COX-2 inhibitor); Other: laboratory biomarker analysis | Completed | NA | |
| NCT01642186 | Study of (1) Everolimus, (2) Estrogen Deprivation Therapy (EDT) With Leuprolide + Letrozole and (3) Everolimus + EDT in Patients with Unresectable Fibrolamellar Hepatocellular Carcinoma (FLL-HCC) | Phase II | n=28; Drug (Target): Everolimus (mTOR inhibitor)/Letrozole (Estrogen inhibitor)/Leuprolide (LHRH/GnRH agonist) | Completed | Fibrolamellar Liver Cancer | [351] |
| NCT01402648 | Estrogen Receptor Beta Agonists (Eviendep) and Polyp Recurrence | Phase I/II | n=60; Dietary Supplement (Target): Eviendep (CM&D Pharma Limited, UK) (ERβ agonist)/Maltodextrins (Placebo) | Completed | Gastrointestinal Neoplasm | NA |
| NCT02089386 | Tamoxifen to Treat Barrett's Metaplasia | Early Phase I | n=7; Drug (Target): Tamoxifen (SERM) | Terminated | NA | |
| NCT02513849 | Tamoxifen in Patients with Oesophageal Cancer | Phase I | n=20 (Estimated); Drug (Target): Tamoxifen (SERM) | Unknown status | NA | |
| NCT00024336 | Radiation Therapy and Tamoxifen in Treating Children with Newly Diagnosed Brain Stem Glioma | Phase II | NA; Drug (Target): Tamoxifen citrate (SERM); Radiation: radiation therapy | Unknown status | Glioblastoma | NA |
| NCT04765098 | Tamoxifen Versus Etoposide After First Recurrence in GBM Patients | Phase II | n=60 (Estimated); Drug (Target): Etoposide (Topoisomerase II inhibitor)/Tamoxifen (SERM) | Recruiting | NA | |
| NCT06501911 | A Study of Bicalutamide with Brain Re-irradiation to Treat Recurrent/Progressive High Grade Glioma | Phase I | n=30 (Estimated); Drug (Target): Bicalutamide (AR inhibitor); Radiation: Intensity-modulated radiation therapy (IMRT) | Not yet recruiting | NA | |
| NCT00004436 | Randomized Study of Hormonal Regulation of Infantile Hemangioma | n/a | n=30; Drug (Target): Leuprolide (LHRH/GnRH agonist)/Prednisone (Glucocorticoid) | Completed | Hemangioma | NA |
| NCT02528643 | A Study to Assess the Efficacy and Safety of Enzalutamide in Subjects with Advanced Hepatocellular Carcinoma | Phase II | n=165; Drug (Target): Enzalutamide (AR inhibitor)/Placebo | Completed | Lung Cancer | [352] |
| NCT00003424 | Tamoxifen in Treating Patients with Primary Liver Cancer | Phase III | n=300 (Estimated); Drug (Target): Tamoxifen citrate (SERM) | Completed | NA | |
| NCT02642939 | Study of Oral Mifepristone as Salvage Therapy in Patients with Advanced or Metastatic Non-Small Cell Lung Cancer | Phase II | n=3; Drug (Target): Mifepristone (GR-II receptor/PR/AR inhibitor) | Terminated | NA | |
| NCT06512207 | A Study on the Efficacy of Androgen Deprivation Therapy Combined with Anti-PD-1 Therapy in Advanced Lung Cancer | n/a | n=80 (Estimated); Drug (Target): Leuprorelin acetate (Androgen inhibitor)/Sintilimab (PD-1 inhibitor) | Recruiting | NA | |
| NCT01556191 | Lung Cancer in Women Treated with Anti-oestrogens anD Inhibitors of EGFR (LADIE) | Phase II | n=379; Drug (Target): Gefitinib (EGFR tyrosine kinase inhibitor)/Fulvestrant (ER inhibitor)/Erlotinib (EGFR tyrosine kinase inhibitor) | Completed | [353] | |
| NCT00592007 | Study Evaluating the Addition of Fulvestrant to Erlotinib in Stage IIIB/IV Non-Small Cell Lung Cancer | Phase II | n=7; Drug (Target): Fulvestrant (ER inhibitor)/Erlotinib (EGFR tyrosine kinase inhibitor) | Terminated | NA | |
| NCT02666105 | Exemestane in Post-Menopausal Women With NSCLC | Phase II | n=6; Drug (Target): Exemestane (Estrogen inhibitor) | Completed | NA | |
| NCT02489123 | Enzalutamide in Treating Patients with Relapsed or Refractory Mantle Cell Lymphoma | Phase II | n=8; Drug (Target): Enzalutamide (AR inhibitor); Other: Laboratory Biomarker Analysis | Terminated | Lymphoma | NA |
| Debio 8200-IMM-101 | A phase I study on the safety and efficacy of triptorelin in combination with nivolumab in men with advanced melanoma resistant to prior anti-PD-1/PD-L1 therapy | Phase I | n=14; Drug (Target): Triptorelin (GnRH agonist)/Nivolumab (PD-1 inhibitor)/Bicalutamide (AR inhibitor) | Completed | Melanoma | [354] |
| NCT00254397 | Melanoma Vaccine with Peptides and Leuprolide | Phase II | n=98; Drug (Target): Leuprolide (LHRH/GnRH agonist); Biological: GP100: 209-217(210M) Peptide/MAGE-3 Peptide | Completed | NA | |
| NCT06320990 | Chemoprevention With Tamoxifen in Pre-Invasive Pancreas Mucinous Cystic Neoplasms Not Undergoing Immediate Resection (MCN_Tam) | Phase I | n=15 (Estimated); Drug (Target): Tamoxifen (SERM) | Not yet recruiting | Pancreatic Neoplasm | NA |
| NCT06222593 | Study to Evaluate the Safety and Efficacy of Bicalutamide in Combination with Sunitinib in Patients with TKIs-resistant RCC | Phase I/II | n=28 (Estimated); Drug (Target): Bicalutamide (Androgen inhibitor)/Sunitinib (CYP3A4 inhibitor) | Not yet recruiting | Renal Cell Cancer | NA |
| NCT02885649 | Enzalutamide Before Surgery in Treating Patients with Kidney Cancer | Early Phase I | n=3; Drug (Target): Enzalutamide (AR inhibitor); Other: Laboratory Biomarker Analysis; Procedure: Nephrectomy | Terminated | NA | |
| NCT03169764 | QUILT-3.047: NANT Head and Neck Squamous Cell Carcinoma (HNSCC) Vaccine: Combination Immunotherapy in Subjects with HNSCC Who Have Progressed on or After Chemotherapy and PD-1/PD-L1 Therapy | Phase I/II | n=0; Drug (Target): Fulvestrant (ER inhibitor) | Withdrawn | Squamous Cell Carcinoma | NA |
| NCT02605863 | Enzalutamide for Bladder Cancer Chemoprevention | Phase II | n=1; Drug (Target): Enzalutamide (AR inhibitor) | Terminated | Urethral bladder cancer | NA |
| NCT02197897 | Evaluation the Treatment of Tamoxifen of Low/Intermediate Risk Bladder Tumors (BCTamoxifen) | Phase II | n=15; Drug (Target): Tamoxifen citrate (SERM) | Completed | NA | |
| NCT06018116 | A Canadian Trial of Bicalutamide in Patients Receiving Maintenance Avelumab for Metastatic Urothelial Cancer. (CANUCK-01) | Phase II | n=0; Drug (Target): Bicalutamide (AR inhibitor)/Placebo | Withdrawn | NA | |
| NCT00710970 | Tamoxifen for Progressive Transitional Cell Carcinoma Following Previous Chemotherapy Treatment | Phase II | n=28; Drug (Target): Tamoxifen (SERM) | Completed | [355], [356] | |
| NCT03197571 | QUILT-3.048: NANT Urothelial Cancer Vaccine: Combination Immunotherapy in Subjects with Urothelial Cancer Who Have Progressed on or After Chemotherapy and PD-1/PD-L1 Therapy | Phase I/II | n=0; Drug (Target): Fulvestrant (ER inhibitor) | Withdrawn | NA |
AR: androgen receptor; COX: cyclooxygenase; EGFR: epidermal growth factor receptor; ER: estrogen receptor; GBM: glioblastoma multiforme; GnRH: gonadotropin-releasing hormone; LHRH: luteinizing hormone-releasing hormone; mTOR: mammalian target of Rapamycin; NSCLC: non-small cell lung cancer; PD-1: programmed death receptor 1; PD-L1: programmed death ligand 1; PR: progesterone receptor; RCC: renal cell carcinoma; SERM: selective estrogen receptor modulator.
Additionally, therapies such as adoptive T cell therapy, immune checkpoint inhibitors, angiogenesis and tyrosine kinase inhibitors, EZH1/2 and BET inhibitors (targeting sex-specific epigenetic and X chromosome inactivation) show sex dimorphism in anti-cancer treatment. Proportion of the treatments are more effective when combined with sex hormone-targeting drugs [317, 318]. Recently, novel cancer drugs with sex bias like telomerase reverse transcriptase (TERT) modulators are emerging, while drugs targeting sex-related genetic factors like telomerase inhibitors still require further investigation [319]. Furthermore, a research revealed treatment biases, highlighting the urgent need for comprehensive sex-based analyses to uncover more clinically significant findings and support the development of personalized, sex-specific cancer treatments [320].
In addition to the previously discussed sex-biased cellular and molecular mechanisms, further investigation is needed to explore sex differences in cancer from the following perspectives: (i) sex-biased cellular senescence in immune cells, as this may play a role in the observed differences in immune-related diseases between sexes [321]. (ii) The impact of sex-specific microbiota on tumor immune responses, since commensal microbiota can produce sex steroids, like androgens, that influence the immune landscape and disease outcomes [322]. (iii) Sex-biased metabolism, which may lead to differences in immune cell function [323]. Notably, significant sex differences in metabolism—such as glycolysis, fatty acid, and bile acid metabolism—have been observed across 13 non-reproductive cancers in the TCGA dataset [323]. (iv) Sex-specific variations in the response to persistent DNA damage signaling, which are critical in managing the tissue-level effects of DNA damage.
In recent research, sex-based differences in the cancer microbiota contributing to cancer progression (colorectal cancer, especially) has been found [324-327], beyond the autoimmunity [328], inflammation [329], or neural and respiratory diseases [330, 331]. In colorectal cancer, the molecular mechanisms of sex-based disparities are focused on sex hormone-gut microbiome axis [324]. Sex-based sexual dimorphism extends to microbe-associated molecular patterns (MAMPs), where estrogens promote greater gut flora diversity, while androgens tend to reduce microbial diversity. This differential impact on the microbiome is associated with cancer development [332]. However, to advance preclinical research and accelerate clinical treatment, challenges remain in fully understanding the role and molecular mechanisms of microbiome-related sexual dimorphisms in cancer. Further investigation is needed to address these gaps.
Understanding sex-biased factors in tumor immunity is crucial for developing sex-specific therapies. In 2016, the U.S. National Institutes of Health (NIH) introduced a policy requiring investigator-initiated grants to consider sex as a biological variable in medical research [322]. Historically, over 60% of immunology studies using animal models did not report the sex of the subjects as of 2009 [333]. However, by 2014, half of the published studies included both sex and age as experimental variables [334]. The significant sex differences observed across all levels of biological organization underscore the fact that findings in males cannot be universally applied to females, and vice versa [333]. Despite this, women remain significantly underrepresented in immunotherapy clinical trials [335], potentially due to concerns that cyclical hormonal changes may influence clinical outcomes [336]. Therefore, further research is needed to understand sex hormone-independent cancer mechanisms. Additionally, sex balance should be carefully considered in clinical trial design before gender-specific targeted therapies are implemented in clinical practice.
Abbreviations
2ME2: 2-Methoxyestradiol
ADT: androgen deprivation therapy
APCs: antigen-presenting cells
AR: androgen receptor
AREs: androgen response elements
ARKO: androgen receptor knockout
BAFF: B-cell activating factor
BMDCs: bone marrow-derived cells
Bregs: regulatory B cells
CAR-T: antigen receptor T cell
CLL: chronic lymphocytic leukemia
CO19ORS: COVID-19-specific immune responses
CpG: cytosine-phosphate-guanine dinucleotide motif
CRPC: castration-resistant prostate cancer
cTECs: cortical thymic epithelial cells
CTLs: cytotoxic T lymphocytes
DCs: dendritic cells
DFS: disease-free survival
DHT: dihydrotestosterone
DLBCL: diffuse large B-cell lymphoma
Dll4: Delta-like 4
E2: 17β-estradiol
EC: endometrial carcinoma
EDY: extreme downregulation of Y-linked genes
EFF: effector T cells
eNOS: endothelial nitric oxide synthase
ERK: extracellular signal-regulated kinase
ER: estrogen receptor
ERα: estrogen receptor alpha
ERβ: estrogen receptor beta
ERE: estrogen response element
ESR1: estrogen receptor 1
ESR2: estrogen receptor 2
GC: Gastric cancer
gMDSCs: granulocytic MDSCs
GPR: G protein-coupled receptor
GR: glucocorticoid receptor
HCC: hepatocellular carcinoma
hCG: human chorionic gonadotropin
HRT: hormone replacement therapy
iAR: intracellular androgen receptors
JNK: c-Jun N-terminal kinase
ICB: immune checkpoint blockade
ICIs: immune checkpoint inhibitors
ICT: immune checkpoint inhibitor therapy
IFN-I: type I interferon
IFN-γ: interferon gamma
ILCs: innate lymphoid cells
KO: knockout
KS: klinefelter syndrome
LOY: Y chromosome mosaic loss
MAMPs: microbe-associated molecular patterns
MAPK: mitogen-activated protein kinase
mAR: membrane androgen receptors
MDSCs: myeloid-derived suppressor cells
MHC: major histocompatibility complex
mMDSCs: mononuclear MDSCs
NE: neutrophil elastase
NETs: neutrophil extracellular traps
NIH: national Institutes of Health
NSCLC: non-small cell lung cancer
OS: overall survival
P4: progesterone
PAAD: pancreatic adenocarcinoma
PD-1: programmed death-1
pDC: plasmacytoid dendritic cell
PEX: progenitor exhausted T cells
PFS: progression-free survival
PI3K: phosphatidylinositol 3 kinase
PIBF: progesterone-induced blocking factor
PKC: protein kinase C
PMNs: polymorphonuclear neutrophils
PPP: pentose phosphate pathway
PR: progesterone receptor
PRL: prolactin
PTC: papillary thyroid carcinoma
RBP: regulatory binding protein
scRNA-seq: Single-cell RNA sequencing
SERMs: selective estrogen receptor modulators
SLE: systemic lupus erythematosus
SupraT: supraphysiologic levels of testosterone
SupraT: supraphysiologic levels of testosterone
T-ALL: T-cell acute lymphoblastic leukemia
TAMs: tumor-associated macrophages
TAN: tumor-associated neutrophil
TCF: T cell factor
TCR: T cell receptor
TEX: terminally exhausted T cells
TERT: telomerase reverse transcriptase
Tfm: testicular feminization
Th: T helper cell
TIL-Bs: Tumor-infiltrating B lymphocytes
TKT: transketolase
TLS: tertiary lymphoid structures
TME: tumor microenvironment
TNBC: triple-negative breast cancer
TNF: tumor necrosis factor
Treg: regulatory T cell
TβRI: transforming growth factor β receptor I
UTX: ubiquitously transcribed tetratricopeptide repeat on chromosome X
VAT: visceral adipose tissue
XCI: X chromosome inactivation
XIST: RNA X-inactivation-specific transcript
Acknowledgements
The figures were generated by Biorender.com. ChatGPT was used for language polishing.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81903135), Sichuan Science and technology Foundation Project, China (Grant No. 2023YFH0079), Youth Fund of Sichuan Provincial People's Hospital (NO. 2017QN09), Open Project of Sichuan Provincial Key Laboratory for Clinical Immunology Translational Medicine (Grant No. LCMYZHYX-KFKT202306).
Authorship contribution statement
Xuerui Tao: Writing - review & editing, Writing - original draft, Conceptualization, Visualization. Yiling Wang: Writing - review & editing, Funding acquisition. Binghua Xiang: Writing - review & editing. Chunmei Hu: Writing - review & editing. Wei Xiong: Writing - review & editing. Wenjun Liao: Writing - review & editing. Shichuan Zhang: Writing - review & editing. Chi Liu: Writing - review & editing. Xiaoxiao Wang: Writing - review & editing, Funding acquisition. Yue Zhao: Writing - review & editing, Supervision, Project administration, Funding acquisition, Conceptualization.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Costa AR, Lança de Oliveira M, Cruz I, Gonçalves I, Cascalheira JF, Santos CRA. The Sex Bias of Cancer. Trends Endocrinol Metab. 2020;31:785-99
2. Zhu Y, Shao X, Wang X, Liu L, Liang H. Sex disparities in cancer. Cancer Lett. 2019;466:35-8
3. Jackson SS, Marks MA, Katki HA, Cook MB, Hyun N, Freedman ND. et al. Sex disparities in the incidence of 21 cancer types: Quantification of the contribution of risk factors. Cancer. 2022;128:3531-40
4. Lungaro L, Costanzini A, Manza F, Barbalinardo M, Gentili D, Guarino M. et al. Impact of Female Gender in Inflammatory Bowel Diseases: A Narrative Review. J Pers Med. 2023;13:165
5. Katz-Greenberg G, Shah S. Sex and Gender Differences in Kidney Transplantation. Semin Nephrol. 2022;42:219-29
6. Baritussio A, Schiavo A, Basso C, Giordani AS, Cheng Cy, Pontara E. et al. Predictors of relapse, death or heart transplantation in myocarditis before the introduction of immunosuppression: negative prognostic impact of female gender, fulminant onset, lower ejection fraction and serum autoantibodies. Eur J Heart Fail. 2022;24:1033-44
7. Maenosono R, Nian Y, Iske J, Liu Y, Minami K, Rommel T. et al. Recipient sex and estradiol levels affect transplant outcomes in an age-specific fashion. Am J Transplant. 2021;21:3239-55
8. Lahita RG. Sex and gender influence on immunity and autoimmunity. Front Immunol. 2023;14:1142723
9. Takahashi T, Iwasaki A. Sex differences in immune responses. Science. 2021;371:347-8
10. Niu P, Li L, Zhang Y, Su Z, Wang B, Liu H. et al. Immune regulation based on sex differences in ischemic stroke pathology. Front Immunol. 2023;14:1087815
11. Yang C, Jin J, Yang Y, Sun H, Wu L, Shen M. et al. Androgen receptor-mediated CD8+ T cell stemness programs drive sex differences in antitumor immunity. Immunity. 2022;55:1268-83 e9
12. Xiao T, Lee J, Gauntner TD, Velegraki M, Lathia JD, Li Z. Hallmarks of sex bias in immuno-oncology: mechanisms and therapeutic implications. Nat Rev Cancer. 2024;24:338-55
13. Ueno T, Horii R, Matsumoto H, Ono M, Maeshima Y, Nitta H. Abstract PO4-01-08: Co-expression of estrogen receptor (ER), progesterone receptor (PR), and Ki67 in a single breast cancer cell indicates a favorable prognosis in ER-positive breast cancer. Cancer Res. 2024;84:PO4-01 -8-PO4-8
14. Koszegi Z, Cheong RY. Targeting the non-classical estrogen pathway in neurodegenerative diseases and brain injury disorders. Front Endocrinol (Lausanne). 2022;13:999236
15. Chhabra A, Tripathi A, Rizvi S, Tyagi RK. Ligand-independent homo-/hetero-dimerization events of ERα and ERβ occur in the cytoplasmic compartment: Evidences from receptor dynamics in live cells. J Steroid Biochem Mol Biol. 2025;247:106668
16. Reid SE, Pantaleo J, Bolivar P, Bocci M, Sjölund J, Morsing M. et al. Cancer-associated fibroblasts rewire the estrogen receptor response in luminal breast cancer, enabling estrogen independence. Oncogene. 2024;43:1113-26
17. Qin C, Lu Y, Zhang H, Zhang Z, Xu W, Wen S. et al. Biological roles and clinical significance of estrogen and androgen receptors in head and neck cancers. J Cancer. 2022;13:2189-99
18. Prossnitz ER, Arterburn JB. International union of basic and clinical pharmacology. XCVII. G protein-coupled estrogen receptor and its pharmacologic modulators. Pharmacol Rev. 2015;67:505-40
19. Arnal JF, Lenfant F, Metivier R, Flouriot G, Henrion D, Adlanmerini M. et al. Membrane and Nuclear Estrogen Receptor Alpha Actions: From Tissue Specificity to Medical Implications. Physiol Rev. 2017;97:1045-87
20. Haupt S, Caramia F, Klein SL, Rubin JB, Haupt Y. Sex disparities matter in cancer development and therapy. Nat Rev Cancer. 2021;21:393-407
21. Tsuchida CA, Brandes N, Bueno R, Trinidad M, Mazumder T, Yu B. et al. Mitigation of chromosome loss in clinical CRISPR-Cas9-engineered T cells. Cell. 2023;186:4567-82.e20
22. Case LK, Wall EH, Dragon JA, Saligrama N, Krementsov DN, Moussawi M. et al. The Y chromosome as a regulatory element shaping immune cell transcriptomes and susceptibility to autoimmune disease. Genome Res. 2013;23:1474-85
23. Mattisson J, Halvardson J, Davies H, Bruhn-Olszewska B, Olszewski P, Danielsson M. et al. Loss of chromosome Y in regulatory T cells. BMC Genomics. 2024;25:243
24. Song CH, Kim N, Nam RH, Choi SI, Jang JY, Kim EH. et al. Ninjurin1 deficiency differentially mitigates colorectal cancer induced by azoxymethane and dextran sulfate sodium in male and female mice. Int J Cancer. 2025;156:826-39
25. Salehi R, Asare-Werehene M, Wyse BA, Abedini A, Pan B, Gutsol A. et al. Granulosa cell-derived miR-379-5p regulates macrophage polarization in polycystic ovarian syndrome. Front Immunol. 2023;14:1104550
26. Zhou WJ, Zhang J, Xie F, Wu JN, Ye JF, Wang J. et al. CD45RO(-)CD8(+) T cell-derived exosomes restrict estrogen-driven endometrial cancer development via the ERβ/miR-765/PLP2/Notch axis. Theranostics. 2021;11:5330-45
27. Yuan B, Clark CA, Wu B, Yang J, Drerup JM, Li T. et al. Estrogen receptor beta signaling in CD8(+) T cells boosts T cell receptor activation and antitumor immunity through a phosphotyrosine switch. J Immunother Cancer. 2021;9:e001932
28. Correction. Estrogen receptor beta signaling in CD8+ T cells boosts T cell receptor activation and antitumor immunity through a phosphotyrosine switch. J Immunother Cancer. 2021;9:e001932
29. Speiser DE, Chijioke O, Schaeuble K, Münz C. CD4+ T cells in cancer. Nat cancer. 2023;4:317-29
30. Reina-Campos M, Scharping NE, Goldrath AW. CD8+ T cell metabolism in infection and cancer. Nat Rev Immunol. 2021;21:718-38
31. Singh V, Nandi S, Ghosh A, Adhikary S, Mukherjee S, Roy S. et al. Epigenetic reprogramming of T cells: unlocking new avenues for cancer immunotherapy. Cancer Metastasis Rev. 2024;43:175-95
32. Wang R, Liu Z, Fan Z, Zhan H. Lipid metabolism reprogramming of CD8+ T cell and therapeutic implications in cancer. Cancer Lett. 2023;567:216267
33. Renaude E, Kroemer M, Borg C, Peixoto P, Hervouet E, Loyon R. et al. Epigenetic reprogramming of CD4+ helper T cells as a strategy to improve anticancer immunotherapy. Front Immunol. 2021;12:669992
34. Biswas SK. Metabolic reprogramming of immune cells in cancer progression. Immunity. 2015;43:435-49
35. Xia L, Oyang L, Lin J, Tan S, Han Y, Wu N. et al. The cancer metabolic reprogramming and immune response. Mol Cancer. 2021;20:1-21
36. Faubert B, Solmonson A, DeBerardinis RJ. Metabolic reprogramming and cancer progression. Science. 2020;368:eaaw5473
37. Katz SG, Rabinovich PM. T Cell Reprogramming Against Cancer. Methods Mol Biol. 2020;2097:3-44
38. Lim AR, Rathmell WK, Rathmell JC. The tumor microenvironment as a metabolic barrier to effector T cells and immunotherapy. Elife. 2020;9:e55185
39. Dunn SE, Perry WA, Klein SL. Mechanisms and consequences of sex differences in immune responses. Nat Rev Nephrol. 2024;20:37-55
40. Klein SL, Flanagan KL. Sex differences in immune responses. Nat Rev Immunol. 2016;16:626-38
41. Shepherd R, Cheung AS, Pang K, Saffery R, Novakovic B. Sexual dimorphism in innate immunity: the role of sex hormones and epigenetics. Front Immunol. 2021;11:604000
42. Silasi M, You Y, Simpson S, Kaislasuo J, Pal L, Guller S. et al. Human chorionic gonadotropin modulates CXCL10 expression through histone methylation in human decidua. Sci Rep. 2020;10:5785
43. Szekeres-Bartho J, Šućurović S, Mulac-Jeričević B. The Role of Extracellular Vesicles and PIBF in Embryo-Maternal Immune-Interactions. Front Immunol. 2018;9:2890
44. Jaillon S, Berthenet K, Garlanda C. Sexual Dimorphism in Innate Immunity. Clin Rev Allergy Immunol. 2019;56:308-21
45. Dauven D, Ehrentraut S, Langwisch S, Zenclussen AC, Schumacher A. Immune Modulatory Effects of Human Chorionic Gonadotropin on Dendritic Cells Supporting Fetal Survival in Murine Pregnancy. Front Endocrinol (Lausanne). 2016;7:146
46. Piccinni MP, Giudizi MG, Biagiotti R, Beloni L, Giannarini L, Sampognaro S. et al. Progesterone favors the development of human T helper cells producing Th2-type cytokines and promotes both IL-4 production and membrane CD30 expression in established Th1 cell clones. J Immunol. 1995;155:128-33
47. Xiao Y, Zheng P, Xu W, Wu Z, Zhang X, Wang R. et al. Progesterone receptor impairs immune respond and down-regulates sensitivity to anti-LAG3 in breast cancer. Transl Res. 2024;271:68-78
48. Krishnan N, R LN, Warrier AV, Yadev I, Anandan J, Sundaram S. et al. Direct DNA binding by BRCA1 on β-hCG promoter and its clinical implications. Heliyon. 2024;10:e37064
49. Vasanthakumar A, Chisanga D, Blume J, Gloury R, Britt K, Henstridge DC. et al. Sex-specific adipose tissue imprinting of regulatory T cells. Nature. 2020;579:581-5
50. Forsyth KS, Jiwrajka N, Lovell CD, Toothacre NE, Anguera MC. The conneXion between sex and immune responses. Nat Rev Immunol. 2024;24:487-502
51. Hogg SJ, Beavis PA, Dawson MA, Johnstone RW. Targeting the epigenetic regulation of antitumour immunity. Nat Rev Drug Discov. 2020;19:776-800
52. Peng Z, Dong X, He M, Zhao Y, Liu Y, Li M. et al. Elevated profiles of peripheral Th22, Th17, Th2 cells, and decreased percentage of Th1 cells in breast cancer patients. Thorac Cancer. 2023;14:3282-94
53. Chakraborty B, Byemerwa J, Krebs T, Lim F, Chang C-Y, McDonnell DP. Estrogen Receptor Signaling in the Immune System. Endocr Rev. 2023;44:117-41
54. Conforti F, Pala L, Bagnardi V, De Pas T, Martinetti M, Viale G. et al. Cancer immunotherapy efficacy and patients' sex: a systematic review and meta-analysis. Lancet Oncol. 2018;19:737-46
55. Adurthi S, Kumar MM, Vinodkumar HS, Mukherjee G, Krishnamurthy H, Acharya KK. et al. Oestrogen Receptor-α binds the FOXP3 promoter and modulates regulatory T-cell function in human cervical cancer. Sci Rep. 2017;7:17289
56. Luo CY, Wang L, Sun C, Li DJ. Estrogen enhances the functions of CD4(+)CD25(+)Foxp3(+) regulatory T cells that suppress osteoclast differentiation and bone resorption in vitro. Cell Mol Immunol. 2011;8:50-8
57. Oh MS, Anker JF, Chae YK. High gene expression of estrogen and progesterone receptors is associated with decreased t cell infiltration in patients with NSCLC. Cancer Treat Res Commun. 2021;27:100317
58. Kwon Y. Potential Pro-Tumorigenic Effect of Bisphenol A in Breast Cancer via Altering the Tumor Microenvironment. Cancers (Basel). 2022;14:3021
59. Xu Y, Huangyang P, Wang Y, Xue L, Devericks E, Nguyen HG. et al. ERα is an RNA-binding protein sustaining tumor cell survival and drug resistance. Cell. 2021;184:5215-29 e17
60. Klinge CM. Estrogenic control of mitochondrial function. Redox Biology. 2020;31:101435
61. Xu Z, Ma T, Zhou J, Gao W, Li Y, Yu S. et al. Nuclear receptor ERRα contributes to castration-resistant growth of prostate cancer via its regulation of intratumoral androgen biosynthesis. Theranostics. 2020;10:4201-16
62. Tao L, Qiu J, Slavin S, Ou Z, Liu Z, Ge J. et al. Recruited T cells promote the bladder cancer metastasis via up-regulation of the estrogen receptor β/IL-1/c-MET signals. Cancer Lett. 2018;430:215-23
63. Zhao L, Huang S, Mei S, Yang Z, Xu L, Zhou N. et al. Pharmacological activation of estrogen receptor beta augments innate immunity to suppress cancer metastasis. Proc Natl Acad Sci U S A. 2018;115:E3673-E81
64. Hoffmann JP, Liu JA, Seddu K, Klein SL. Sex hormone signaling and regulation of immune function. Immunity. 2023;56:2472-91
65. Tong D. Selective estrogen receptor modulators contribute to prostate cancer treatment by regulating the tumor immune microenvironment. J Immunother Cancer. 2022;10:e002944
66. Jefferi NES, Shamhari AA, Azhar NKZN, Shin JGY, Kharir NAM, Azhar MA. et al. The Role of ERα and ERβ in Castration-Resistant Prostate Cancer and Current Therapeutic Approaches. Biomedicines. 2023;11:826
67. Song S, Tang H, Quan W, Shang A, Ling C. Estradiol initiates the immune escape of non-small cell lung cancer cells via ERβ/SIRT1/FOXO3a/PD-L1 axis. Int Immunopharmacol. 2022;107:108629
68. Guo D, Liu X, Zeng C, Cheng L, Song G, Hou X. et al. Estrogen receptor β activation ameliorates DSS-induced chronic colitis by inhibiting inflammation and promoting Treg differentiation. Int Immunopharmacol. 2019;77:105971
69. Wang T, Jin J, Qian C, Lou J, Lin J, Xu A. et al. Estrogen/ER in anti-tumor immunity regulation to tumor cell and tumor microenvironment. Cancer Cell Int. 2021;21:1-13
70. Goodman WA, Bedoyan SM, Havran HL, Richardson B, Cameron MJ, Pizarro TT. Impaired estrogen signaling underlies regulatory T cell loss-of-function in the chronically inflamed intestine. Proc Natl Acad Sci U S A. 2020;117:17166-76
71. Cipolletti M, Bartoloni S, Busonero C, Parente M, Leone S, Acconcia F. A New Anti-Estrogen Discovery Platform Identifies FDA-Approved Imidazole Anti-Fungal Drugs as Bioactive Compounds against ERα Expressing Breast Cancer Cells. Int J Mol Sci. 2021;22:2915
72. Cao M, Wei J, Pan Y, Wang L, Li Z, Hu Y. et al. Antagonistic mechanisms of bisphenol analogues on the estrogen receptor α in zebrafish embryos: Experimental and computational studies. Sci Total Environ. 2023;857:159259
73. Kajihara N, Ge Y, Seino K-i. Blocking of oestrogen signals improves anti-tumour effect regardless of oestrogen receptor alpha expression in cancer cells. Br J Cancer. 2023;129:935-46
74. Sahu A, Wang X, Munson P, Klomp JP, Wang X, Gu SS. et al. Discovery of targets for immune-metabolic antitumor drugs identifies estrogen-related receptor alpha. Cancer Discov. 2023;13:672-701
75. Tinivella A, Nwachukwu JC, Angeli A, Foschi F, Benatti AL, Pinzi L. et al. Design, synthesis, biological evaluation and crystal structure determination of dual modulators of carbonic anhydrases and estrogen receptors. Eur J Med Chem. 2023;246:115011
76. Goldberg J, Qiao N, Guerriero JL, Gross B, Meneksedag Y, Lu YF. et al. Estrogen Receptor Mutations as Novel Targets for Immunotherapy in Metastatic Estrogen Receptor-positive Breast Cancer. Cancer Res Commun. 2024;4:496-504
77. Xiong S, Song K, Xiang H, Luo G. Dual-target inhibitors based on ERα: Novel therapeutic approaches for endocrine resistant breast cancer. Eur J Med Chem. 2024;270:116393
78. Shuai C, Yang X, Pan H, Han W. Estrogen Receptor Downregulates Expression of PD-1/PD-L1 and Infiltration of CD8+ T Cells by Inhibiting IL-17 Signaling Transduction in Breast Cancer. Front Oncol. 2020;10:582863
79. Rubin JB, Lagas JS, Broestl L, Sponagel J, Rockwell N, Rhee G. et al. Sex differences in cancer mechanisms. Biol Sex Differ. 2020;11:17
80. Walecki M, Eisel F, Klug J, Baal N, Paradowska-Dogan A, Wahle E. et al. Androgen receptor modulates Foxp3 expression in CD4+CD25+Foxp3+ regulatory T-cells. Mol Biol Cell. 2015;26:2845-57
81. Kissick HT, Sanda MG, Dunn LK, Pellegrini KL, On ST, Noel JK. et al. Androgens alter T-cell immunity by inhibiting T-helper 1 differentiation. Proc Natl Acad Sci U S A. 2014;111:9887-92
82. Mauvais-Jarvis F, Lange CA, Levin ER. Membrane-Initiated Estrogen, Androgen, and Progesterone Receptor Signaling in Health and Disease. Endocr Rev. 2021;43:720-42
83. Henze L, Schwinge D, Schramm C. The Effects of Androgens on T Cells: Clues to Female Predominance in Autoimmune Liver Diseases? Front Immunol. 2020;11:1567
84. Zhang X, Cheng L, Gao C, Chen J, Liao S, Zheng Y. et al. Androgen Signaling Contributes to Sex Differences in Cancer by Inhibiting NF-κB Activation in T Cells and Suppressing Antitumor Immunity. Cancer Res. 2023;83:906-21
85. Lee J, Yurkovetskiy LA, Reiman D, Frommer L, Strong Z, Chang A. et al. Androgens contribute to sex bias of autoimmunity in mice by T cell-intrinsic regulation of Ptpn22 phosphatase expression. Nat Commun. 2024;15:7688
86. Zhang MA, Rego D, Moshkova M, Kebir H, Chruscinski A, Nguyen H. et al. Peroxisome proliferator-activated receptor (PPAR)α and -γ regulate IFNγ and IL-17A production by human T cells in a sex-specific way. Proc Natl Acad Sci U S A. 2012;109:9505-10
87. Sorrentino C, Musiani P, Pompa P, Cipollone G, Di Carlo E. Androgen deprivation boosts prostatic infiltration of cytotoxic and regulatory T lymphocytes and has no effect on disease-free survival in prostate cancer patients. Clin Cancer Res. 2011;17:1571-81
88. Perniola R. Twenty Years of AIRE. Front Immunol. 2018;9:98
89. Guan X, Polesso F, Wang C, Sehrawat A, Hawkins RM, Murray SE. et al. Androgen receptor activity in T cells limits checkpoint blockade efficacy. Nature. 2022;606:791-6
90. Qu LG, Wardan H, Davis ID, Pezaro C, Sluka P. Effects of estrogen receptor signaling on prostate cancer carcinogenesis. Transl Res. 2020;222:56-66
91. Erzurumlu Y, Dogan HK, Catakli D, Aydogdu E, Muhammed MT. Estrogens drive the endoplasmic reticulum-associated degradation and promote proto-oncogene c-Myc expression in prostate cancer cells by androgen receptor/estrogen receptor signaling. J Cell Commun Signal. 2023;17:793-811
92. Muñoz-Rojas AR, Mathis D. Tissue regulatory T cells: regulatory chameleons. Nat Rev Immunol. 2021;21:597-611
93. Belk JA, Daniel B, Satpathy AT. Epigenetic regulation of T cell exhaustion. Nat Immunol. 2022;23:848-60
94. Lee J, Nicosia M, Hong ES, Silver DJ, Li C, Bayik D. et al. Sex-Biased T-cell Exhaustion Drives Differential Immune Responses in Glioblastoma. Cancer Discov. 2023;13:2090-105
95. Androgen Receptor Signaling Reduces Male Antitumor CD8+ T-cell Activity. Cancer Discov. 2022;12:1836
96. Kwon H, Schafer JM, Song NJ, Kaneko S, Li A, Xiao T. et al. Androgen conspires with the CD8(+) T cell exhaustion program and contributes to sex bias in cancer. Sci Immunol. 2022;7:eabq2630
97. Zhang L, Romero P. Metabolic Control of CD8(+) T Cell Fate Decisions and Antitumor Immunity. Trends Mol Med. 2018;24:30-48
98. Chen J, Huang C-P, Quan C, Zu X, Ou Z, Tsai Y-C. et al. The androgen receptor in bladder cancer. Nat Rev Urol. 2023;20:560-74
99. Xiao T, Schafer J, Song N-j, Wang C, Weltge P, Li X. et al. Novel mechanisms of androgen receptor-centered transcriptional regulatory network in regulating CD8 +T cell exhaustion and sex bias in cancer. J Immunol. 2023;210:171.13-13
100. Quatrini L, Ricci B, Ciancaglini C, Tumino N, Moretta L. Regulation of the Immune System Development by Glucocorticoids and Sex Hormones. Front Immunol. 2021;12:672853
101. Velardi E, Tsai JJ, Holland AM, Wertheimer T, Yu VW, Zakrzewski JL. et al. Sex steroid blockade enhances thymopoiesis by modulating Notch signaling. J Exp Med. 2014;211:2341-9
102. Zhu M-L, Bakhru P, Conley B, Nelson JS, Free M, Martin A. et al. Sex bias in CNS autoimmune disease mediated by androgen control of autoimmune regulator. Nat commun. 2016;7:11350
103. Dragin N, Bismuth J, Cizeron-Clairac G, Biferi MG, Berthault C, Serraf A. et al. Estrogen-mediated downregulation of AIRE influences sexual dimorphism in autoimmune diseases. J Clin Invest. 2016;126:1525-37
104. Youness A, Cenac C, Faz-López B, Grunenwald S, Barrat FJ, Chaumeil J. et al. TLR8 escapes X chromosome inactivation in human monocytes and CD4+ T cells. Biol Sex Differ. 2023;14:60
105. Miquel C-H, Faz-Lopez B, Guéry J-C. Influence of X chromosome in sex-biased autoimmune diseases. J Autoimmun. 2023;137:102992
106. Ma D, Ma J, Zhao C, Tai W. Reasons why women are more likely to develop primary biliary cholangitis. Heliyon. 2024;10:e25634
107. Cui X, Zhang C, Xu Z, Wang S, Li X, Stringer-Reasor E. et al. Dual CRISPR interference and activation for targeted reactivation of X-linked endogenous FOXP3 in human breast cancer cells. Mol Cancer. 2022;21:38
108. Glasner A, Plitas G. Tumor resident regulatory T cells. Semin Immunol. 2021;52:101476
109. Itoh Y, Golden LC, Itoh N, Matsukawa MA, Ren E, Tse V. et al. The X-linked histone demethylase Kdm6a in CD4+ T lymphocytes modulates autoimmunity. J Clin Invest. 2019;129:3852-63
110. Lee J, Lathia JD. Skull bones harbour immune cells that are poised to target brain tumours. Nature. 2024;633:528-9
111. Mitchell JE, Lund MM, Starmer J, Ge K, Magnuson T, Shpargel KB. et al. UTX promotes CD8(+) T cell-mediated antiviral defenses but reduces T cell durability. Cell Rep. 2021;35:108966
112. Collier JL, Weiss SA, Pauken KE, Sen DR, Sharpe AH. Not-so-opposite ends of the spectrum: CD8+ T cell dysfunction across chronic infection, cancer and autoimmunity. Nat Immunol. 2021;22:809-19
113. Hatzioannou A, Boumpas A, Papadopoulou M, Papafragkos I, Varveri A, Alissafi T. et al. Regulatory T Cells in Autoimmunity and Cancer: A Duplicitous Lifestyle. Front Immunol. 2021;12:731947
114. Maruhashi T, Sugiura D, Okazaki I-m, Shimizu K, Maeda TK, Ikubo J. et al. Binding of LAG-3 to stable peptide-MHC class II limits T cell function and suppresses autoimmunity and anti-cancer immunity. Immunity. 2022;55:912-24 e8
115. Crowther MD, Sewell AK. The burgeoning role of MR1-restricted T-cells in infection, cancer and autoimmune disease. Curr Opin Immunol. 2021;69:10-7
116. Zitvogel L, Perreault C, Finn OJ, Kroemer G. Beneficial autoimmunity improves cancer prognosis. Nat Rev Clin Oncol. 2021;18:591-602
117. Dumanski JP, Halvardson J, Davies H, Rychlicka-Buniowska E, Mattisson J, Moghadam BT. et al. Immune cells lacking Y chromosome show dysregulation of autosomal gene expression. Cell Mol Life Sci. 2021;78:4019-33
118. Abdel-Hafiz HA, Schafer JM, Chen X, Xiao T, Gauntner TD, Li Z. et al. Y chromosome loss in cancer drives growth by evasion of adaptive immunity. Nature. 2023;619:624-31
119. Yang J, Xu J, Wang W, Zhang B, Yu X, Shi S. Epigenetic regulation in the tumor microenvironment: molecular mechanisms and therapeutic targets. Signal Transduct Target Ther. 2023;8:210
120. Potaczek DP, Harb H, Michel S, Alhamwe BA, Renz H, Tost J. Epigenetics and Allergy: from Basic Mechanisms to Clinical Applications. Epigenomics. 2017;9:539-71
121. Migliore L, Nicolì V, Stoccoro A. Gender Specific Differences in Disease Susceptibility: The Role of Epigenetics. Biomedicines. 2021;9:652
122. Liu Q, Zhu F, Liu X, Lu Y, Yao K, Tian N. et al. Non-oxidative pentose phosphate pathway controls regulatory T cell function by integrating metabolism and epigenetics. Nat Metab. 2022;4:559-74
123. Salumets A, Tserel L, Rumm AP, Türk L, Kingo K, Saks K. et al. Epigenetic quantification of immunosenescent CD8+ TEMRA cells in human blood. Aging Cell. 2022;21:e13607
124. Shi A, Lin C, Wang J, Zhong J, Lyu J. EPRIM: An approach of identifying cancer immune-related epigenetic regulators. Mol Ther Nucleic Acids. 2024;35:102100
125. R P, Rakshit S, Shanmugam G, George M, Sarkar K. Wiskott Aldrich syndrome protein (WASp)-deficient Th1 cells promote R-loop-driven transcriptional insufficiency and transcription-coupled nucleotide excision repair factor (TC-NER)-driven genome-instability in the pathogenesis of T cell acute lymphoblastic leukemia. Clin Immunol. 2024;263:110204
126. Garcia-Prat M, Batlle-Masó L, Parra-Martínez A, Franco-Jarava C, Martinez-Gallo M, Aguiló-Cucurull A. et al. Role of Skewed X-Chromosome Inactivation in Common Variable Immunodeficiency. J Clin Immunol. 2024;44:54
127. Fan H, Zhao G, Ren D, Liu F, Dong G, Hou Y. Gender differences of B cell signature related to estrogen-induced IFI44L/BAFF in systemic lupus erythematosus. Immunol Lett. 2017;181:71-8
128. Souyris M, Cenac C, Azar P, Daviaud D, Canivet A, Grunenwald S. et al. TLR7 escapes X chromosome inactivation in immune cells. Sci Immunol. 2018;3:eaap8855
129. Kroeger DR, Milne K, Nelson BH. Tumor-infiltrating plasma cells are associated with tertiary lymphoid structures, cytolytic T-cell responses, and superior prognosis in ovarian cancer. Clin Cancer Res. 2016;22:3005-15
130. Downs-Canner SM, Meier J, Vincent BG, Serody JS. B Cell Function in the Tumor Microenvironment. Annu Rev Immunol. 2022;40:169-93
131. Garaud S, Buisseret L, Solinas C, Gu-Trantien C, de Wind A, Van den Eynden G. et al. Tumor-infiltrating B cells signal functional humoral immune responses in breast cancer. JCI insight. 2019;4:e129641
132. Shen P, Fillatreau S. Antibody-independent functions of B cells: a focus on cytokines. Nat Rev Immunol. 2015;15:441-51
133. Castino GF, Cortese N, Capretti G, Serio S, Di Caro G, Mineri R. et al. Spatial distribution of B cells predicts prognosis in human pancreatic adenocarcinoma. Oncoimmunology. 2016;5:e1085147
134. Engelhard V, Conejo-Garcia JR, Ahmed R, Nelson BH, Willard-Gallo K, Bruno TC. et al. B cells and cancer. Cancer Cell. 2021;39:1293-6
135. Shu DH, Ho WJ, Kagohara LT, Girgis A, Shin SM, Danilova L. et al. Immunotherapy response induces divergent tertiary lymphoid structure morphologies in hepatocellular carcinoma. Nat Immunol. 2024;25:2110-23
136. Chaurio RA, Anadon CM, Lee Costich T, Payne KK, Biswas S, Harro CM. et al. TGF-β-mediated silencing of genomic organizer SATB1 promotes Tfh cell differentiation and formation of intra-tumoral tertiary lymphoid structures. Immunity. 2022;55:115-28.e9
137. Lam BM, Verrill C. Clinical Significance of Tumour-Infiltrating B Lymphocytes (TIL-Bs) in Breast Cancer: A Systematic Literature Review. Cancers (Basel). 2023;15:1164
138. Laumont CM, Banville AC, Gilardi M, Hollern DP, Nelson BH. Tumour-infiltrating B cells: immunological mechanisms, clinical impact and therapeutic opportunities. Nat Rev Cancer. 2022;22:414-30
139. Li YY, Li SJ, Liu MC, Chen Z, Li L, Shen F. et al. B cells and tertiary lymphoid structures are associated with survival in papillary thyroid cancer. J Endocrinol Invest. 2023;46:2247-56
140. Zou X, Liu Y, Lin X, Wang R, Dai Z, Chen Y. et al. Characterization of Estrogen Receptors in Pancreatic Adenocarcinoma with Tertiary Lymphoid Structures. Cancers (Basel). 2023;15:828
141. Pashos CL, Flowers CR, Kay NE, Weiss M, Lamanna N, Farber C. et al. Association of health-related quality of life with gender in patients with B-cell chronic lymphocytic leukemia. Support Care Cancer. 2013;21:2853-60
142. Langendonk M, de Jong MRW, Smit N, Seiler J, Reitsma B, Ammatuna E. et al. Identification of the estrogen receptor beta as a possible new tamoxifen-sensitive target in diffuse large B-cell lymphoma. Blood Cancer J. 2022;12:36
143. Warakomska M, Tynecka M, Lemancewicz D, Grubczak K, Dzieciol J, Moniuszko M. et al. The effects of BAFF and APRIL signaling on non-small cell lung cancer cell proliferation and invasiveness. Oncol Lett. 2021;22:1-8
144. Liu W, Stachura P, Xu HC, Váraljai R, Shinde P, Ganesh NU. et al. BAFF attenuates immunosuppressive monocytes in the melanoma tumor microenvironment. Cancer Res. 2022;82:264-77
145. Ullah MA, Mackay F. The BAFF-APRIL system in cancer. Cancers (Basel). 2023;15:1791
146. Aziz K, Munjal K, Howe K, Yarchoan M. B-cell activating factor (BAFF) enhances antitumor immunity. Cancer Res. 2024;84:3978 -
147. Wong DP, Roy NK, Zhang K, Anukanth A, Asthana A, Shirkey-Son NJ. et al. A BAFF ligand-based CAR-T cell targeting three receptors and multiple B cell cancers. Nat Commun. 2022;13:217
148. Lv Z, Wang T-y, Bi Y, Li D, Wu Q, Wang B. et al. BAFF overexpression in triple-negative breast cancer promotes tumor growth by inducing IL-10-secreting regulatory B cells that suppress anti-tumor T cell responses. Breast Cancer Res Treat. 2024;209:405-418
149. Ladikou E-E, Kassi E. The emerging role of estrogen in B cell malignancies. Leuk Lymphoma. 2017;58:528-39
150. Garcia-Martinez L, Zhang Y, Nakata Y, Chan HL, Morey L. Epigenetic mechanisms in breast cancer therapy and resistance. Nat Commun. 2021;12:1786
151. Yohannan B, George B. B-lymphoid blast phase-chronic myeloid leukemia: current therapeutics. Int J Mol Sci. 2022;23:11836
152. Santana-Sánchez P, Vaquero-García R, Legorreta-Haquet MV, Chávez-Sánchez L, Chávez-Rueda AK. Hormones and B-cell development in health and autoimmunity. Front Immunol. 2024;15:1385501
153. Hasselblom S, Ridell B, Nilsson-Ehle H, Andersson PO. The impact of gender, age and patient selection on prognosis and outcome in diffuse large B-cell lymphoma - a population-based study. Leuk Lymphoma. 2007;48:736-45
154. Liu B, Zhou M, Li X, Zhang X, Wang Q, Liu L. et al. Interrogation of gender disparity uncovers androgen receptor as the transcriptional activator for oncogenic miR-125b in gastric cancer. Cell Death Dis. 2021;12:441
155. Doshi B, Athans SR, Woloszynska A. Biological differences underlying sex and gender disparities in bladder cancer: current synopsis and future directions. Oncogenesis. 2023;12:44
156. Olsen NJ, Gu X, Kovacs WJ. Bone marrow stromal cells mediate androgenic suppression of B lymphocyte development. J Clin Invest. 2001;108:1697-704
157. Zhao R, Chen X, Ma W, Zhang J, Guo J, Zhong X. et al. A GPR174-CCL21 module imparts sexual dimorphism to humoral immunity. Nature. 2020;577:416-20
158. Morelli C, Chiodo C, Nocito MC, Cormace A, Catalano S, Sisci D. et al. Androgens Modulate Bcl-2 Agonist of Cell Death (BAD) Expression and Function in Breast Cancer Cells. Int J Mol Sci. 2023;24:13464
159. Issa J-PJ, Zehnbauer BA, Civin CI, Collector MI, Sharkis SJ, Davidson NE. et al. The estrogen receptor CpG island is methylated in most hematopoietic neoplasms. Cancer Res. 1996;56:973-7
160. Brait M, Banerjee M, Maldonado L, Ooki A, Loyo M, Guida E. et al. Promoter methylation of MCAM, ERα and ERβ in serum of early stage prostate cancer patients. Oncotarget. 2017;8:15431
161. Roma A, Spagnuolo PA. Estrogen receptors alpha and beta in acute myeloid leukemia. Cancers (Basel). 2020;12:907
162. Sukocheva OA, Lukina E, Friedemann M, Menschikowski M, Hagelgans A, Aliev G. The crucial role of epigenetic regulation in breast cancer anti-estrogen resistance: Current findings and future perspectives. Semin Cancer Biol. 2022;82:35-59
163. Riihijarvi S, Taskinen M, Jerkeman M, Leppa S. Male gender is an adverse prognostic factor in B-cell lymphoma patients treated with immunochemotherapy. Eur J Haematol. 2011;86:124-8
164. Wang L, Zhang S, Xin J. Sex differences in abdominal visceral fat composition and metabolism as predictors of DLBCL prognosis: A retrospective cohort study. Eur J Radiol. 2024;170:111205
165. Nilsson N, Carlsten H. Estrogen induces suppression of natural killer cell cytotoxicity and augmentation of polyclonal B cell activation. Cell Immunol. 1994;158:131-9
166. Dahut M, Fousek K, Horn LA, Angstadt S, Qin H, Hamilton DH. et al. Fulvestrant increases the susceptibility of enzalutamide-resistant prostate cancer cells to NK-mediated lysis. J Immunother Cancer. 2023;11:e007386
167. Wu Q, Tian P, He D, Jia Z, He Y, Luo W. et al. SCUBE2 mediates bone metastasis of luminal breast cancer by modulating immune-suppressive osteoblastic niches. Cell Res. 2023;33:464-78
168. Curran EM, Berghaus LJ, Vernetti NJ, Saporita AJ, Lubahn DB, Estes DM. Natural Killer Cells Express Estrogen Receptor-α and Estrogen Receptor-β and Can Respond to Estrogen Via a Non-Estrogen Receptor-α-Mediated Pathway. Cell Immunol. 2001;214:12-20
169. Carbonnel M, Daclin C, Tarantino N, Groiseau O, Morin V, Rousseau A. et al. Plasticity of natural killer cells in pregnant patients infected with SARS-CoV-2 and their neonates during childbirth. Front Immunol. 2022;13:893450
170. Wolfson B, Padget MR, Schlom J, Hodge JW. Exploiting off-target effects of estrogen deprivation to sensitize estrogen receptor negative breast cancer to immune killing. J Immunother Cancer. 2021;9:e002258
171. Godoy-Pacheco A, García-Chagollán M, Ramírez-De-Arellano A, Hernández-Silva CD, Villegas-Pineda JC, Ramírez-López IG. et al. Differential modulation of natural killer cell cytotoxicity by 17β-estradiol and prolactin through the NKG2D/NKG2DL axis in cervical cancer cells. Oncol Lett. 2022;24:1-12
172. Guerrero B, Hassouneh F, Delgado E, Casado JG, Tarazona R. Natural killer cells in recurrent miscarriage: An overview. J Reprod Immunol. 2020;142:103209
173. Alvarez M, Simonetta F, Baker J, Morrison AR, Wenokur AS, Pierini A. et al. Indirect Impact of PD-1/PD-L1 Blockade on a Murine Model of NK Cell Exhaustion. Front Immunol. 2020;11:7
174. Liu Q, You B, Meng J, Huang C-P, Dong G, Wang R. et al. Targeting the androgen receptor to enhance NK cell killing efficacy in bladder cancer by modulating ADAR2/circ_0001005/PD-L1 signaling. Cancer Gene Ther. 2022;29:1988-2000
175. Zhang W, Xu C, Yang Z, Zhou J, Peng W, Zhang X. et al. Circular RNAs in tumor immunity and immunotherapy. Mol Cancer. 2024;23:171
176. Tang M, Sun Y, Huang C-P, Chen L, Liu B, You B. et al. High dose androgen suppresses natural killer cytotoxicity of castration-resistant prostate cancer cells via altering AR/circFKBP5/miRNA-513a-5p/PD-L1 signals. Cell Death Dis. 2022;13:746
177. Shi L, Lin H, Li G, Jin R-A, Xu J, Sun Y. et al. Targeting androgen receptor (AR)→ IL12A signal enhances efficacy of sorafenib plus NK cells immunotherapy to better suppress HCC progression. Mol Cancer Ther. 2016;15:731-42
178. Cheng MI, Li JH, Riggan L, Chen B, Tafti RY, Chin S. et al. The X-linked epigenetic regulator UTX controls NK cell-intrinsic sex differences. Nat Immunol. 2023;24:780-91
179. Galaras A, Verykokakis M. UT(se)X differences during immune responses. Nat Immunol. 2023;24:738-40
180. Rubin JB, Abou-Antoun T, Ippolito JE, Llaci L, Marquez CT, Wong JP. et al. Epigenetic developmental mechanisms underlying sex differences in cancer. J Clin Invest. 2024;134:e180071
181. Reid Cahn A, Farkas AM. XXtra killer cells. Nat Rev Immunol. 2022;22:532 -
182. Cáceres A, Jene A, Esko T, Pérez-Jurado LA, González JR. Extreme Downregulation of Chromosome Y and Cancer Risk in Men. J Natl Cancer Inst. 2020;112:913-20
183. Bruhn-Olszewska B, Markljung E, Rychlicka-Buniowska E, Sarkisyan D, Filipowicz N, Dumanski JP. The effects of loss of Y chromosome on male health. Nat Rev Genet. 2025
184. Xia M, Wang B, Wang Z, Zhang X, Wang X. Epigenetic regulation of NK cell-mediated antitumor immunity. Front Immunol. 2021;12:672328
185. Su S, Hua D, Li J-P, Zhang X-N, Bai L, Cao L-B. et al. Modulation of innate immune response to viruses including SARS-CoV-2 by progesterone. Signal Transduct Target Ther. 2022;7:137
186. Griesbeck M, Ziegler S, Laffont S, Smith N, Chauveau L, Tomezsko P. et al. Sex differences in plasmacytoid dendritic cell levels of IRF5 drive higher IFN-α production in women. J Immunol. 2015;195:5327-36
187. Ziegler SM, Beisel C, Sutter K, Griesbeck M, Hildebrandt H, Hagen SH. et al. Human pDCs display sex-specific differences in type I interferon subtypes and interferon α/β receptor expression. Eur J Immunol. 2017;47:251-6
188. Asano T, Boisson B, Onodi F, Matuozzo D, Moncada-Velez M, Maglorius Renkilaraj MRL. et al. X-linked recessive TLR7 deficiency in ~1% of men under 60 years old with life-threatening COVID-19. Sci Immunol. 2021;6:eabl4348
189. Webb K, Peckham H, Radziszewska A, Menon M, Oliveri P, Simpson F. et al. Sex and pubertal differences in the type 1 interferon pathway associate with both X chromosome number and serum sex hormone concentration. Front Immunol. 2019;9:3167
190. Spiering AE, de Vries TJ. Why Females Do Better: The X Chromosomal TLR7 Gene-Dose Effect in COVID-19. Front Immunol. 2021;12:756262
191. Sciarra F, Campolo F, Franceschini E, Carlomagno F, Venneri MA. Gender-Specific Impact of Sex Hormones on the Immune System. Int J Mol Sci. 2023;24:6302
192. Krueger K, Lamenza F, Gu H, El-Hodiri H, Wester J, Oberdick J. et al. Sex differences in susceptibility to substance use disorder: Role for X chromosome inactivation and escape? Mol Cell Neurosci. 2023;125:103859
193. Wang JP, Zhang L, Madera RF, Woda M, Libraty DH. Plasmacytoid dendritic cell interferon-α production to R-848 stimulation is decreased in male infants. BMC Immunol. 2012;13:1-5
194. Sampson OL, Jay C, Adland E, Csala A, Lim N, Ebbrecht SM. et al. Gonadal androgens are associated with decreased type I interferon production by plasmacytoid dendritic cells and increased IgG titres to BNT162b2 following co-vaccination with live attenuated influenza vaccine in adolescents. Front Immunol. 2024;15:1329805
195. Grünhagel B, Borggrewe M, Hagen SH, Ziegler SM, Henseling F, Glau L. et al. Reduction of IFN-I responses by plasmacytoid dendritic cells in a longitudinal trans men cohort. iScience. 2023;26:108209
196. Koh YT, Gray A, Higgins SA, Hubby B, Kast WM. Androgen ablation augments prostate cancer vaccine immunogenicity only when applied after immunization. The Prostate. 2009;69:571-84
197. Corrales JJ, Almeida M, Cordero M, Martín-Martín L, Méndez C, Miralles JM. et al. Enhanced immunological response by dendritic cells in male hypogonadism. Eur J Clin Invest. 2012;42:1205-12
198. Hepworth MR, Hardman MJ, Grencis RK. The role of sex hormones in the development of Th2 immunity in a gender-biased model of Trichuris muris infection. Eur J Immunol. 2010;40:406-16
199. Stojić-Vukanić Z, Pilipović I, Bufan B, Stojanović M, Leposavić G. Age and sex determine CD4+ T cell stimulatory and polarizing capacity of rat splenic dendritic cells. Biogerontology. 2020;21:83-107
200. Xia HJ, Zhang GH, Wang RR, Zheng YT. The influence of age and sex on the cell counts of peripheral blood leukocyte subpopulations in Chinese rhesus macaques. Cell Mol Immunol. 2009;6:433-40
201. Ben-Batalla I, Vargas-Delgado ME, von Amsberg G, Janning M, Loges S. Influence of Androgens on Immunity to Self and Foreign: Effects on Immunity and Cancer. Front Immunol. 2020;11:1184
202. Hatzidaki E, Papasotiriou I. Personalized dendritic cell vaccination in cancer therapy: An evidence-based research study. J Cancer Res Ther. 2023;19:S52-S58
203. He G, Zhang H, Zhou J, Wang B, Chen Y, Kong Y. et al. Peritumoural neutrophils negatively regulate adaptive immunity via the PD-L1/PD-1 signalling pathway in hepatocellular carcinoma. J Exp Clin Cancer Res. 2015;34:141
204. Prossnitz ER, Barton M. The G protein-coupled oestrogen receptor GPER in health and disease: an update. Nat Rev Endocrinol. 2023;19:407-24
205. Gupta S, Nakabo S, Blanco LP, O'Neil LJ, Wigerblad G, Goel RR. et al. Sex differences in neutrophil biology modulate response to type I interferons and immunometabolism. Proc Natl Acad Sci U S A. 2020;117:16481-91
206. Cramer DW, Vitonis AF. Signatures of reproductive events on blood counts and biomarkers of inflammation: Implications for chronic disease risk. PLoS One. 2017;12:e0172530
207. Habib P, Dreymueller D, Roesing B, Botung H, Slowik A, Zendedel A. et al. Estrogen serum concentration affects blood immune cell composition and polarization in human females under controlled ovarian stimulation. J Steroid Biochem Mol Biol. 2018;178:340-7
208. Sthoeger Z, Chiorazzi N, Lahita R. Regulation of the immune response by sex hormones. I. In vitro effects of estradiol and testosterone on pokeweed mitogen-induced human B cell differentiation. J Immunol. 1988;141:91-8
209. Aslanian-Kalkhoran L, Mehdizadeh A, Aghebati-Maleki L, Danaii S, Shahmohammadi-Farid S, Yousefi M. The role of neutrophils and neutrophil extracellular traps (NETs) in stages, outcomes and pregnancy complications. J Reprod Immunol. 2024;163:104237
210. Patel D, Dodd WS, Lucke-Wold B, Chowdhury MAB, Hosaka K, Hoh BL. Neutrophils: Novel Contributors to Estrogen-Dependent Intracranial Aneurysm Rupture Via Neutrophil Extracellular Traps. J Am Heart Assoc. 2023;12:e029917
211. Chatterjee S. Reliability of sexual dimorphism in blood. Indian J Physiol Pharmacol. 2014;58:400-2
212. Chung HH, Or YZ, Shrestha S, Loh JT, Lim CL, Ong Z. et al. Estrogen reprograms the activity of neutrophils to foster protumoral microenvironment during mammary involution. Sci Rep. 2017;7:46485
213. Song W, Li L, He D, Xie H, Chen J, Yeh C-R. et al. Infiltrating neutrophils promote renal cell carcinoma (RCC) proliferation via modulating androgen receptor (AR)→ c-Myc signals. Cancer Lett. 2015;368:71-8
214. Clausen F, Behrens H-M, Krüger S, Röcken C. Sexual dimorphism in gastric cancer: tumor-associated neutrophils predict patient outcome only for women. J Cancer Res Clin Oncol. 2020;146:53-66
215. Quaas A, Pamuk A, Klein S, Quantius J, Rehkaemper J, Barutcu AG. et al. Sex-specific prognostic effect of CD66b-positive tumor-infiltrating neutrophils (TANs) in gastric and esophageal adenocarcinoma. Gastric Cancer. 2021;24:1213-26
216. Markman JL, Porritt RA, Wakita D, Lane ME, Martinon D, Noval Rivas M. et al. Loss of testosterone impairs anti-tumor neutrophil function. Nat Commun. 2020;11:1613
217. Hreha TN, Collins CA, Cole EB, Jin RJ, Hunstad DA. Androgen exposure impairs neutrophil maturation and function within the infected kidney. Mbio. 2024;15:e03170-23
218. Scheff NN, Bhattacharya A, Dowse E, Dang RX, Dolan JC, Wang S. et al. Neutrophil-mediated endogenous analgesia contributes to sex differences in oral cancer pain. Front Integr Neurosci. 2018;12:52
219. Alsamraae M, Costanzo-Garvey D, Teply BA, Boyle S, Sommerville G, Herbert ZT. et al. Androgen receptor inhibition suppresses anti-tumor neutrophil response against bone metastatic prostate cancer via regulation of TβRI expression. Cancer Lett. 2023;579:216468
220. Jensen SS, Christensen E, Petersen MJ, Munther R, Hansen A, Bak M. et al. Systemic TLR7/8 micelles trigger a novel and potent anti-tumor response by strong recruitment of neutrophils leading to massive tumor cell killing. J Clin Oncol. 2022;40:2576 -
221. Lu J, Zhong X, Guo C, Tang L, Yu N, Peng C. et al. TLR7-MyD88-DC-CXCL16 axis results neutrophil activation to elicit inflammatory response in pustular psoriasis. Cell Death Dis. 2023;14:315
222. Gao C, Qiao T, Yuan S, Zhuang X. The Preliminary study for Postoperative radiotherapy survival associated with RUNX3 and TLR9 expression in lung cancer. Cancer Manag Res. 2021;13:4497-507
223. Kou M, Lu W, Zhu M, Qu K, Wang L, Yu Y. Massively recruited sTLR9+ neutrophils in rapidly formed nodules at the site of tumor cell inoculation and their contribution to a pro-tumor microenvironment. Cancer Immunol Immunother. 2023;72:2671-86
224. Yao L, Sheng X, Dong X, Zhou W, Li Y, Ma X. et al. Neutrophil extracellular traps mediate TLR9/Merlin axis to resist ferroptosis and promote triple negative breast cancer progression. Apoptosis. 2023;28:1484-95
225. Ma M, Ghosh S, Tavernari D, Katarkar A, Clocchiatti A, Mazzeo L. et al. Sustained androgen receptor signaling is a determinant of melanoma cell growth potential and tumorigenesis. J Exp Med. 2020;218:e20201137
226. Yamaguchi M, Takagi K, Sato M, Sato A, Miki Y, Onodera Y. et al. Androgens enhance the ability of intratumoral macrophages to promote breast cancer progression. Oncol Rep. 2021;46:188
227. Izumi K, Fang LY, Mizokami A, Namiki M, Li L, Lin WJ. et al. Targeting the androgen receptor with siRNA promotes prostate cancer metastasis through enhanced macrophage recruitment via CCL2/CCR2-induced STAT3 activation. EMBO Mol Med. 2013;5:1383-401 -401
228. Cioni B, Zaalberg A, van Beijnum JR, Melis MHM, van Burgsteden J, Muraro MJ. et al. Androgen receptor signalling in macrophages promotes TREM-1-mediated prostate cancer cell line migration and invasion. Nat Commun. 2020;11:4498
229. Zhang LJ, Xiong Y, Nilubol N, He M, Bommareddi S, Zhu X. et al. Testosterone regulates thyroid cancer progression by modifying tumor suppressor genes and tumor immunity. Carcinogenesis. 2015;36:420-8
230. Kumar R, Mendonca J, Owoyemi O, Boyapati K, Thomas N, Kanacharoen S. et al. Supraphysiologic Testosterone Induces Ferroptosis and Activates Immune Pathways through Nucleophagy in Prostate Cancer. Cancer Res. 2021;81:5948-62
231. Lee GT, Kim JH, Kwon SJ, Stein MN, Hong JH, Nagaya N. et al. Dihydrotestosterone Increases Cytotoxic Activity of Macrophages on Prostate Cancer Cells via TRAIL. Endocrinology. 2019;160:2049-60
232. Rubin JB. The spectrum of sex differences in cancer. Trends Cancer. 2022;8:303-15
233. Rae JM, Lippman ME. The role of estrogen receptor signaling in suppressing the immune response to cancer. J Clin Invest. 2021;131:e115476
234. McGuinness C, Britt KL. Estrogen receptor regulation of the immune microenvironment in breast cancer. J Steroid Biochem Mol Biol. 2024;240:106517
235. Tower H, Dall G, Davey A, Stewart M, Lanteri P, Ruppert M. et al. Estrogen-induced immune changes within the normal mammary gland. Sci Rep. 2022;12:18986
236. Ciucci A, Zannoni GF, Buttarelli M, Lisi L, Travaglia D, Martinelli E. et al. Multiple direct and indirect mechanisms drive estrogen-induced tumor growth in high grade serous ovarian cancers. Oncotarget. 2016;7:8155-71
237. Ning C, Xie B, Zhang L, Li C, Shan W, Yang B. et al. Infiltrating Macrophages Induce ERα Expression through an IL17A-mediated Epigenetic Mechanism to Sensitize Endometrial Cancer Cells to Estrogen. Cancer Res. 2016;76:1354-66
238. Svensson S, Abrahamsson A, Rodriguez GV, Olsson A-K, Jensen L, Cao Y. et al. CCL2 and CCL5 Are Novel Therapeutic Targets for Estrogen-Dependent Breast Cancer. Clin Cancer Res. 2015;21:3794-805
239. Yang W, Lu Y, Xu Y, Xu L, Zheng W, Wu Y. et al. Estrogen represses hepatocellular carcinoma (HCC) growth via inhibiting alternative activation of tumor-associated macrophages (TAMs). J Biol Chem. 2012;287:40140-9
240. Tong H, Ke J-Q, Jiang F-Z, Wang X-J, Wang F-Y, Li Y-R. et al. Tumor-associated macrophage-derived CXCL8 could induce ERα suppression via HOXB13 in endometrial cancer. Cancer Lett. 2016;376:127-36
241. He M, Yu W, Chang C, Miyamoto H, Liu X, Jiang K. et al. Estrogen receptor α promotes lung cancer cell invasion via increase of and cross-talk with infiltrated macrophages through the CCL2/CCR2/MMP9 and CXCL12/CXCR4 signaling pathways. Mol Oncol. 2020;14:1779-99
242. Hollmén M, Roudnicky F, Karaman S, Detmar M. Characterization of macrophage - cancer cell crosstalk in estrogen receptor positive and triple-negative breast cancer. Sci Rep. 2015;5:9188
243. Sousa S, Brion R, Lintunen M, Kronqvist P, Sandholm J, Mönkkönen J. et al. Human breast cancer cells educate macrophages toward the M2 activation status. Breast Cancer Res. 2015;17:1-14
244. Nahmias Blank D, Hermano E, Sonnenblick A, Maimon O, Rubinstein AM, Drai E. et al. Macrophages upregulate estrogen receptor expression in the model of obesity-associated breast carcinoma. Cells. 2022;11:2844
245. Chen Y-C, Young M-J, Chang H-P, Liu C-Y, Lee C-C, Tseng Y-L. et al. Estradiol-mediated inhibition of DNMT1 decreases p53 expression to induce M2-macrophage polarization in lung cancer progression. Oncogenesis. 2022;11:25
246. Rothenberger NJ, Somasundaram A, Stabile LP. The Role of the Estrogen Pathway in the Tumor Microenvironment. Int J Mol Sci. 2018;19:611
247. Wu S-Y, Sharma S, Wu K, Tyagi A, Zhao D, Deshpande RP. et al. Tamoxifen suppresses brain metastasis of estrogen receptor-deficient breast cancer by skewing microglia polarization and enhancing their immune functions. Breast Cancer Res. 2021;23:35
248. Deswal B, Bagchi U, Santra MK, Garg M, Kapoor S. Inhibition of STAT3 by 2-Methoxyestradiol suppresses M2 polarization and protumoral functions of macrophages in breast cancer. BMC Cancer. 2024;24:1129
249. Kobatake K, Ikeda K-i, Nakata Y, Yamasaki N, Ueda T, Kanai A. et al. Kdm6a Deficiency Activates Inflammatory Pathways, Promotes M2 Macrophage Polarization, and Causes Bladder Cancer in Cooperation with p53 Dysfunction. Clin Cancer Res. 2020;26:2065-79
250. Deny M, Arroba Nuñez LA, Romano M, Denis O, Casimir G, Chamekh M. Sex difference in innate inflammatory response and macrophage polarization in Streptococcus agalactiae-induced pneumonia and potential role of microRNA-223-3p. Sci Rep. 2022;12:17126
251. Zając AE, Czarnecka AM, Rutkowski P. The Role of Macrophages in Sarcoma Tumor Microenvironment and Treatment. Cancers (Basel). 2023;15:5294
252. Horitani K, Chavkin NW, Arai Y, Wang Y, Ogawa H, Yura Y. et al. Disruption of the Uty epigenetic regulator locus in hematopoietic cells phenocopies the profibrotic attributes of Y chromosome loss in heart failure. Nat Cardiovasc Res. 2024;3:343-355
253. Lasser SA, Ozbay Kurt FG, Arkhypov I, Utikal J, Umansky V. Myeloid-derived suppressor cells in cancer and cancer therapy. Nat Rev Clin Oncol. 2024;21:147-64
254. Ren J, Hou Y, Wang T. Roles of estrogens on myeloid-derived suppressor cells in cancer and autoimmune diseases. Cell Mol Immunol. 2018;15:724-6
255. Bayik D, Zhou Y, Park C, Hong C, Vail D, Silver DJ. et al. Myeloid-Derived Suppressor Cell Subsets Drive Glioblastoma Growth in a Sex-Specific Manner. Cancer Discov. 2020;10:1210-25
256. De Velasco MA, Kura Y, Sakai K, Saito Y, Minami T, Yoshimura K. et al. Abstract 104: Profiling peripheral blood to predict response to targeted androgen receptor axis therapy in mice. Cancer Res. 2024;84:104 -
257. Muralidhar A, Gamat-Huber M, Vakkalanka S, McNeel DG. Sequence of androgen receptor-targeted vaccination with androgen deprivation therapy affects anti-prostate tumor efficacy. J Immunother Cancer. 2024;12:e008848
258. Kadel S, Ainsua-Enrich E, Hatipoglu I, Turner S, Singh S, Khan S. et al. A major population of functional KLRG1-ILC2s in female lungs contributes to a sex bias in ILC2 numbers. Immunohorizons. 2018;2:74-86
259. Blanquart E, Laffont S, Guéry J-C. Sex hormone regulation of innate lymphoid cells. Biomed J. 2021;44:144-56
260. Cephus J-Y, Stier MT, Fuseini H, Yung JA, Toki S, Bloodworth MH. et al. Testosterone attenuates group 2 innate lymphoid cell-mediated airway inflammation. Cell Rep. 2017;21:2487-99
261. Warren KJ, Sweeter JM, Pavlik JA, Nelson AJ, Devasure JM, Dickinson JD. et al. Sex differences in activation of lung-related type 2 innate lymphoid cells in experimental asthma. Ann Allergy, Asthma Immunol. 2017;118:233-4
262. Zychlinsky Scharff A, Rousseau M, Lacerda Mariano L, Canton T, Consiglio CR, Albert ML. et al. Sex differences in IL-17 contribute to chronicity in male versus female urinary tract infection. JCI Insight. 2019;5:e122998
263. Seillet C, Jacquelot N. Sensing of physiological regulators by innate lymphoid cells. Cell Mol Immunol. 2019;16:442-51
264. Samarasinghe AE, Woolard SN, Boyd KL, Hoselton SA, Schuh JM, McCullers JA. The immune profile associated with acute allergic asthma accelerates clearance of influenza virus. Immunol Cell Biol. 2014;92:449-59
265. Chi L, Liu C, Gribonika I, Gschwend J, Corral D, Han S-J. et al. Sexual dimorphism in skin immunity is mediated by an androgen-ILC2-dendritic cell axis. Science. 2024;384:eadk6200
266. Mackey E, Moeser AJ. Sex differences in mast cell-associated disorders: a life span perspective. Cold Spring Harb Perspect Biol. 2022;14:a039172
267. McNeel DG, Gardner TA, Higano CS, Kantoff PW, Small EJ, Wener MH. et al. A Transient Increase in Eosinophils Is Associated with Prolonged Survival in Men with Metastatic Castration-Resistant Prostate Cancer Who Receive Sipuleucel-T. Cancer Immunol Res. 2014;2:988-99
268. Artham S, Chang C-Y, McDonnell DP. Eosinophilia in cancer and its regulation by sex hormones. Trends Endocrinol Metab. 2023;34:5-20
269. McDonnell DP, Wardell SE, Chang CY, Norris JD. Next-Generation Endocrine Therapies for Breast Cancer. J Clin Oncol. 2021;39:1383-8
270. Tamaki M, Konno Y, Kobayashi Y, Takeda M, Itoga M, Moritoki Y. et al. Expression and functional roles of G-protein-coupled estrogen receptor (GPER) in human eosinophils. Immunol Lett. 2014;160:72-8
271. Kaune T, Griesmann H, Theuerkorn K, Hämmerle M, Laumen H, Krug S. et al. Gender-specific changes of the gut microbiome correlate with tumor development in murine models of pancreatic cancer. iScience. 2023;26:106841
272. Lakshmikanth T, Consiglio C, Sardh F, Forlin R, Wang J, Tan Z. et al. Immune system adaptation during gender-affirming testosterone treatment. Nature. 2024;633:155-64
273. Li H, Jiang W, Liu S, Yang M, Chen S, Pan Y. et al. Connecting the mechanisms of tumor sex differences with cancer therapy. Mol Cell Biochem. 2024;479:213-31
274. Özdemir BC, Dotto GP. Sex Hormones and Anticancer Immunity. Clin Cancer Res. 2019;25:4603-10
275. Allegra A, Caserta S, Genovese S, Pioggia G, Gangemi S. Gender Differences in Oxidative Stress in Relation to Cancer Susceptibility and Survival. Antioxidants. 2023;12:1255
276. Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262
277. Schonfeld SJ, Pfeiffer RM, Lacey JV Jr, Berrington de González A, Doody MM, Greenlee RT. et al. Hormone-related Risk Factors and Postmenopausal Breast Cancer Among Nulliparous Versus Parous Women: An Aggregated Study. Am J Epidemiol. 2011;173:509-17
278. Li C, Fan Z, Lin X, Cao M, Song F, Song F. Parity and risk of developing breast cancer according to tumor subtype: A systematic review and meta-analysis. Cancer Epidemiol. 2021;75:102050
279. Karakas LA, Atilgan AO, Akilli H, Kuscu UE, Haberal A, Ayhan A. Nulliparity and postmenopausal status are independent factors of malignancy potential of endometrial intraepithelial neoplasia in polyps. Int J Gynaecol Obstet. 2021;152:433-8
280. Mao X, Omeogu C, Karanth S, Joshi A, Meernik C, Wilson L. et al. Association of reproductive risk factors and breast cancer molecular subtypes: a systematic review and meta-analysis. BMC Cancer. 2023;23:644
281. Kumar N, Ehsan S, Banerjee S, Fernandez Perez C, Lhuilier I, Neuner J. et al. The unique risk factor profile of triple-negative breast cancer: a comprehensive meta-analysis. J Natl Cancer Inst. 2024;116:1210-9
282. Gleicher N. Why are reproductive cancers more common in nulliparous women? Reprod Biomed Online. 2013;26:416-9
283. Cereser B, Yiu A, Tabassum N, Del Bel Belluz L, Zagorac S, Ancheta KRZ. et al. The mutational landscape of the adult healthy parous and nulliparous human breast. Nat Commun. 2023;14:5136
284. Dunneram Y, Greenwood DC, Cade JE. Diet, menopause and the risk of ovarian, endometrial and breast cancer. Proc Nutr Soc. 2019;78:438-48
285. Chung H-F, Gete DG, Mishra GD. Age at menopause and risk of lung cancer: A systematic review and meta-analysis. Maturitas. 2021;153:1-10
286. Franceschi S, Gallus S, Talamini R, Tavani A, Negri E, Vecchia CL. Menopause and colorectal cancer. Br J Cancer. 2000;82:1860-2
287. Amant F, von Minckwitz G, Han SN, Bontenbal M, Ring AE, Giermek J. et al. Prognosis of women with primary breast cancer diagnosed during pregnancy: results from an international collaborative study. J Clin Oncol. 2013;31:2532-9
288. Sutherland CM, Loutfi A, Mather FJ, Carter RD, Krementz ET. Effect of pregnancy upon malignant melanoma. Surg Gynecol Obstet. 1983;157:443-6
289. Einstein MH, Levine NF, Nevadunsky NS. Menopause and Cancers. Endocrinol Metab Clin North Am. 2015;44:603-17
290. Rodriguez-Lara V, Hernandez-Martinez JM, Arrieta O. Influence of estrogen in non-small cell lung cancer and its clinical implications. J Thorac Dis. 2018;10:482-97
291. Chen W, Liu H, Huang X, Qian L, Chen L, Zhou Y. et al. A single-cell landscape of pre- and post-menopausal high-grade serous ovarian cancer ascites. iScience. 2023;26:107712
292. Jang Y-C, Leung CY, Huang H-L. Association of hormone replacement therapy with risk of gastric cancer: a systematic review and meta-analysis. Sci Rep. 2022;12:12997
293. Collaborative Group on Epidemiological Studies of Ovarian C. Menopausal hormone use and ovarian cancer risk: individual participant meta-analysis of 52 epidemiological studies. Lancet. 2015;385:1835-42
294. Type and timing of menopausal hormone therapy and breast cancer risk. individual participant meta-analysis of the worldwide epidemiological evidence. Lancet. 2019;394:1159-68
295. Lagergren K, Lagergren J, Brusselaers N. Hormone replacement therapy and oral contraceptives and risk of oesophageal adenocarcinoma: a systematic review and meta-analysis. Int J Cancer. 2014;135:2183-90
296. Jang Y-C, Huang H-L, Leung CY. Association of hormone replacement therapy with mortality in colorectal cancer survivor: a systematic review and meta-analysis. BMC Cancer. 2019;19:1199
297. Narod SA. Hormone replacement therapy and the risk of breast cancer. Nat Rev Clin Oncol. 2011;8:669-76
298. Qu K, Zaba LC, Giresi PG, Li R, Longmire M, Kim YH. et al. Individuality and variation of personal regulomes in primary human T cells. Cell Syst. 2015;1:51-61
299. e Sousa LBdA, Jonkers I, Syx L, Dunkel I, Chaumeil J, Picard C. et al. Kinetics of Xist-induced gene silencing can be predicted from combinations of epigenetic and genomic features. Genome Res. 2019;29:1087-99
300. Delbridge AR, Kueh AJ, Ke F, Zamudio NM, El-Saafin F, Jansz N. et al. Loss of p53 causes stochastic aberrant X-chromosome inactivation and female-specific neural tube defects. Cell Rep. 2019;27:442-54 e5
301. Chamekh M, Casimir G. Editorial: Sexual Dimorphism of the Immune Inflammatory Response in Infectious and Non-infectious Diseases. Front Immunol. 2019;10:107
302. Spolarics Z, Peña G, Qin Y, Donnelly RJ, Livingston DH. Inherent X-linked genetic variability and cellular mosaicism unique to females contribute to sex-related differences in the innate immune response. Front Immunol. 2017;8:1455
303. Hollows R, Wei W, Cazier JB, Mehanna H, Parry G, Halford G. et al. Association between loss of Y chromosome and poor prognosis in male head and neck squamous cell carcinoma. Head Neck. 2019;41:993-1006
304. Naugler WE, Sakurai T, Kim S, Maeda S, Kim K, Elsharkawy AM. et al. Gender Disparity in Liver Cancer Due to Sex Differences in MyD88-Dependent IL-6 Production. Science. 2007;317:121-4
305. Caetano MS, Hassane M, Van HT, Bugarin E, Cumpian AM, McDowell CL. et al. Sex specific function of epithelial STAT3 signaling in pathogenesis of K-ras mutant lung cancer. Nat Commun. 2018;9:4589
306. Hewagama A, Patel D, Yarlagadda S, Strickland FM, Richardson BC. Stronger inflammatory/cytotoxic T-cell response in women identified by microarray analysis. Genes Immun. 2009;10:509-16
307. Kovats S. Estrogen receptors regulate innate immune cells and signaling pathways. Cell Immunol. 2015;294:63-9
308. Laffont S, Seillet C, Guéry J-C. Estrogen Receptor-Dependent Regulation of Dendritic Cell Development and Function. Front Immunol. 2017;8:108
309. Kim JY, Casaccia-Bonnefil P. Interplay of hormones and p53 in modulating gender dimorphism of subventricular zone cell number. J Neurosci Res. 2009;87:3297-305
310. La Vecchia S, Fontana S, Salaroglio IC, Anobile DP, Digiovanni S, Akman M. et al. Increasing membrane polyunsaturated fatty acids sensitizes non-small cell lung cancer to anti-PD-1/PD-L1 immunotherapy. Cancer Lett. 2024;604:217221
311. Reizel Y, Spiro A, Sabag O, Skversky Y, Hecht M, Keshet I. et al. Gender-specific postnatal demethylation and establishment of epigenetic memory. Genes Dev. 2015;29:923-33
312. Bramble MS, Roach L, Lipson A, Vashist N, Eskin A, Ngun T. et al. Sex-specific effects of testosterone on the sexually dimorphic transcriptome and epigenome of embryonic neural stem/progenitor cells. Sci Rep. 2016;6:36916
313. Irelli A, Sirufo MM, D'Ugo C, Ginaldi L, De Martinis M. Sex and Gender Influences on Cancer Immunotherapy Response. Biomedicines. 2020;8:232
314. Sieveking DP, Lim P, Chow RW, Dunn LL, Bao S, McGrath KC. et al. A sex-specific role for androgens in angiogenesis. J Exp Med. 2010;207:345-52
315. Lu Q, Schnitzler GR, Ueda K, Iyer LK, Diomede OI, Andrade T. et al. ER alpha rapid signaling is required for estrogen induced proliferation and migration of vascular endothelial cells. PLoS One. 2016;11:e0152807
316. Likhite N, Yadav V, Milliman EJ, Sopariwala DH, Lorca S, Narayana NP. et al. Loss of estrogen-related receptor alpha facilitates angiogenesis in endothelial cells. Mol Cell Biol. 2019;39:e00411-18
317. Pala L, De Pas T, Catania C, Giaccone G, Mantovani A, Minucci S. et al. Sex and cancer immunotherapy: Current understanding and challenges. Cancer Cell. 2022;40:695-700
318. Dai C, Dehm SM, Sharifi N. Targeting the Androgen Signaling Axis in Prostate Cancer. J Clin Oncol. 2023;41:4267-78
319. El Zarif T, Machaalani M, Nawfal R, Nassar AH, Xie W, Choueiri TK. et al. TERT Promoter Mutations Frequency Across Race, Sex, and Cancer Type. The Oncologist. 2023;29:8-14
320. Kammula AV, Schäffer AA, Rajagopal PS, Kurzrock R, Ruppin E. Outcome differences by sex in oncology clinical trials. Nat Commun. 2024;15:2608
321. Marquez EJ, Chung CH, Marches R, Rossi RJ, Nehar-Belaid D, Eroglu A. et al. Sexual-dimorphism in human immune system aging. Nat Commun. 2020;11:751
322. Pernigoni N, Zagato E, Calcinotto A, Troiani M, Mestre RP, Cali B. et al. Commensal bacteria promote endocrine resistance in prostate cancer through androgen biosynthesis. Science. 2021;374:216-24
323. Yuan Y, Liu L, Chen H, Wang Y, Xu Y, Mao H. et al. Comprehensive Characterization of Molecular Differences in Cancer between Male and Female Patients. Cancer Cell. 2016;29:711-22
324. Wu Z, Huang Y, Zhang R, Zheng C, You F, Wang M. et al. Sex differences in colorectal cancer: with a focus on sex hormone-gut microbiome axis. Cell Commun Signal. 2024;22:167
325. Liao H, Li C, Ai Y, Kou Y. Gut microbiome is more stable in males than in females during the development of colorectal cancer. J Appl Microbiol. 2021;131:435-48
326. Khan ZA, Ghorbani M, Heffinger L, Damdimopoulos A, Moro CF, Bjornstedt M. et al. Genderized Gut and Oral Microbiome Shifts: Uncovering Sex-Specific Dysbiosis in Pancreatic Cancer. bioRxiv. 2024. 2024 10. 02.616338
327. Koti M, Ingersoll MA, Gupta S, Lam CM, Li X, Kamat AM. et al. Sex differences in bladder cancer immunobiology and outcomes: a collaborative review with implications for treatment. Eur Urol Oncol. 2020;3:622-30
328. Yurkovetskiy L, Burrows M, Khan AA, Graham L, Volchkov P, Becker L. et al. Gender bias in autoimmunity is influenced by microbiota. Immunity. 2013;39:400-12
329. Del Pinto R, Ferri C, Giannoni M, Cominelli F, Pizarro TT, Pietropaoli D. Meta-analysis of oral microbiome reveals sex-based diversity in biofilms during periodontitis. JCI insight. 2024;9:e171311
330. El-Ansary A, Bhat RS, Zayed N. Gut microbiome and sex bias in autism spectrum disorders. Curr Behav Neurosci Rep. 2020;7:22-31
331. Beauruelle C, Guilloux C-A, Lamoureux C, Héry-Arnaud G. The human microbiome, an emerging key-player in the sex gap in respiratory diseases. Front Med (Lausanne). 2021;8:600879
332. Rizzetto L, Fava F, Tuohy KM, Selmi C. Connecting the immune system, systemic chronic inflammation and the gut microbiome: The role of sex. J Autoimmun. 2018;92:12-34
333. Beery AK, Zucker I. Sex bias in neuroscience and biomedical research. Neurosci Biobehav Rev. 2011;35:565-72
334. Florez-Vargas O, Brass A, Karystianis G, Bramhall M, Stevens R, Cruickshank S. et al. Bias in the reporting of sex and age in biomedical research on mouse models. Elife. 2016;5:e13615
335. Onkar SS, Carleton NM, Lucas PC, Bruno TC, Lee AV, Vignali DAA. et al. The Great Immune Escape: Understanding the Divergent Immune Response in Breast Cancer Subtypes. Cancer Discov. 2023;13:23-40
336. He F, Furones AR, Landegren N, Fuxe J, Sarhan D. Sex dimorphism in the tumor microenvironment - From bench to bedside and back. Semin Cancer Biol. 2022;86:166-79
337. Evans ET, Horst B, Arend RC, Mythreye K. Evolving roles of activins and inhibins in ovarian cancer pathophysiology. Am J Physiol Cell Physiol. 2023;324:C428-c37
338. Pinjusic K, Dubey OA, Egorova O, Nassiri S, Meylan E, Faget J. et al. Activin-A impairs CD8 T cell-mediated immunity and immune checkpoint therapy response in melanoma. J Immunother Cancer. 2022;10:e004533
339. Cai Y, Shen X, Lu L, Yan H, Huang H, Gaule P. et al. Bile acid distributions, sex-specificity, and prognosis in colorectal cancer. Biol Sex Differ. 2022;13:61
340. Pilipović I, Stojić-Vukanić Z, Leposavić G. Adrenoceptors as potential target for add-on immunomodulatory therapy in multiple sclerosis. Pharmacol Ther. 2023;243:108358
341. Rømer AMA, Thorseth M-L, Madsen DH. Immune Modulatory Properties of Collagen in Cancer. Front Immunol. 2021;12:791453
342. Buendía-González FO, Legorreta-Herrera M. The Similarities and Differences between the Effects of Testosterone and DHEA on the Innate and Adaptive Immune Response. Biomolecules. 2022;12:1768
343. Tulchiner G, Pichler R, Ulmer H, Staudacher N, Lindner AK, Brunner A. et al. Sex-specific hormone changes during immunotherapy and its influence on survival in metastatic renal cell carcinoma. Cancer Immunol Immunother. 2021;70:2805-17
344. Juriol L, Valeff N, Dibo M, Ventimiglia MS, Correale J, Jensen F. Human chorionic gonadotropin regulates cytokine production by lymphocytes from patients with multiple sclerosis. J Reprod Immunol. 2024;164:104280
345. Lam CSP, Cheng S, Choong K, Larson MG, Murabito JM, Newton-Cheh C. et al. Influence of Sex and Hormone Status on Circulating Natriuretic Peptides. J Am Coll Cardiol. 2011;58:618-26
346. Mezzasoma L, Talesa VN, Costanzi E, Bellezza I. Natriuretic Peptides Regulate Prostate Cells Inflammatory Behavior: Potential Novel Anticancer Agents for Prostate Cancer. Biomolecules. 2021;11:794
347. Li T, Jiang Y-H, Wang X, Hou D, Jia S-W, Wang Y-F. Immune-regulating effect of oxytocin and its association with the hypothalamic-pituitary axes. J Neuroimmunol. 2024;394:578419
348. Borba V, Carrera-Bastos P, Zandman-Goddard G, Lucia A, Shoenfeld Y. Prolactin's paradox: Friend, foe, or both in immune regulation? Autoimmun Rev. 2024;23:103643
349. Dumais KM, Veenema AH. Vasopressin and oxytocin receptor systems in the brain: Sex differences and sex-specific regulation of social behavior. Front Neuroendocrinol. 2016;40:1-23
350. Mou R, Ma J, Ju X, Wu Y, Chen Q, Li J. et al. Vasopressin drives aberrant myeloid differentiation of hematopoietic stem cells, contributing to depression in mice. Cell Stem Cell. 2024;31:1794-812.e10
351. Lim II, Farber BA LaQuaglia MP. Advances in fibrolamellar hepatocellular carcinoma: a review. Eur J Pediatr Surg. 2014;24:461-6
352. Ryoo B-Y, Palmer DH, Park SR, Rimassa L, Debashis Sarker, Daniele B. et al. Efficacy and Safety Results from a Phase 2, Randomized, Double-Blind Study of Enzalutamide Versus Placebo in Advanced Hepatocellular Carcinoma. Clin Drug Investig. 2021;41:795-808
353. Mazieres J, Barlesi F, Rouquette I, Molinier O, Besse B, Monnet I. et al. Randomized Phase II Trial Evaluating Treatment with EGFR-TKI Associated with Antiestrogen in Women with Nonsquamous Advanced-Stage NSCLC: IFCT-1003 LADIE Trial. Clin Cancer Res. 2020;26:3172-81
354. Robert C, Lebbé C, Lesimple T, Lundström E, Nicolas V, Gavillet B. et al. Phase I Study of Androgen Deprivation Therapy in Combination with Anti-PD-1 in Melanoma Patients Pretreated with Anti-PD-1. Clin Cancer Res. 2023;29:858-65
355. Shen SS, Smith CL, Hsieh J-T, Yu J, Kim IY, Jian W. et al. Expression of estrogen receptors-α and -β in bladder cancer cell lines and human bladder tumor tissue. Cancer. 2006;106:2610-6
356. Kim HT, Kim BC, Kim IY, Mamura M, Seong DH, Jang JJ. et al. Raloxifene, a mixed estrogen agonist/antagonist, induces apoptosis through cleavage of BAD in TSU-PR1 human cancer cells. J Biol Chem. 2002;277:32510-5
Author contact
Corresponding authors: Yue Zhao, zhaoyueyeahcom; Xiaoxiao Wang, wangxiaoxiaouestc.edu.cn; Chi Liu, liuchi_1230com.
 Global reach, higher impact
Global reach, higher impact