13.3
Impact Factor
Theranostics 2024; 14(5):1815-1828. doi:10.7150/thno.89701 This issue Cite
Research Paper
Precision peptide theranostics: developing N- to C-terminus optimized theranostics targeting cholecystokinin-2 receptor
1. Department of Radiopharmaceutical Sciences, Cancer Imaging, The Peter MacCallum Cancer Centre, Victoria 3000, Australia.
2. Sir Peter MacCallum Department of Oncology, The University of Melbourne, Victoria 3010, Australia.
3. Cancer Imaging, Peter MacCallum Cancer Centre, Melbourne, Victoria 3000, Australia.
4. Models of Cancer Translational Research Centre, The Peter MacCallum Cancer Centre, Victoria 3000, Australia.
5. The Bio21 Institute, School of Chemistry, The University of Melbourne, Melbourne, Victoria, 3010 Australia.
6. Medicinal Chemistry, Monash Institute of Pharmaceutical Sciences, Faculty of Pharmacy and Pharmaceutical Sciences, Monash University (Parkville Campus), Parkville, Victoria 3052, Australia.
Abstract
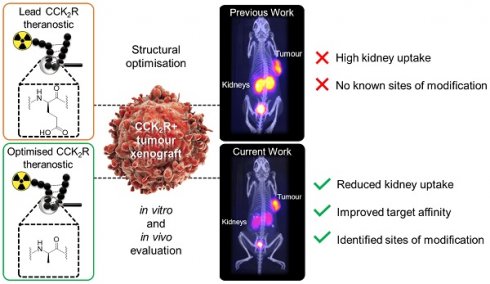
Peptides are ideal for theranostic development as they afford rapid target accumulation, fast clearance from background tissue, and exhibit good tissue penetration. Previously, we developed a novel series of peptides that presented discreet folding propensity leading to an optimal candidate [68Ga]Ga-DOTA-GA1 ([D-Glu]6-Ala-Tyr-NMeGly-Trp-NMeNle-Asp-Nal-NH2) with 50 pM binding affinity against cholecystokinin-2 receptors (CCK2R). However, we were confronted with challenges of unfavorably high renal uptake.
Methods: A structure activity relationship study was undertaken of the lead theranostic candidate. Prudent structural modifications were made to the peptide scaffold to evaluate the contributions of specific N-terminal residues to the overall biological activity. Optimal candidates were then evaluated in nude mice bearing transfected A431-CCK2 tumors, and their biodistribution was quantitated ex vivo.
Results: We identified and confirmed that D-Glu3 to D-Ala3 substitution produced 2 optimal candidates, [68Ga]Ga-DOTA-GA12 and [68Ga]Ga-DOTA-GA13. These radiopeptides presented with high target/background ratios, enhanced tumor retention, excellent metabolic stability in plasma and mice organ homogenates, and a 4-fold reduction in renal uptake, significantly outperforming their non-alanine counterparts.
Conclusions: Our study identified novel radiopharmaceutical candidates that target the CCK2R. Their high tumor uptake and reduced renal accumulation warrant clinical translation.
 Global reach, higher impact
Global reach, higher impact