13.3
Impact Factor
Theranostics 2024; 14(4):1683-1700. doi:10.7150/thno.89805 This issue Cite
Research Paper
Cancer-associated fibroblasts reprogram cysteine metabolism to increase tumor resistance to ferroptosis in pancreatic cancer
1. Department of Radiology, The First Affiliated Hospital of Xiamen University, 55 Zhenhai Road, Siming District, Xiamen, Fujian Province, People's Republic of China.
2. Ningbo University Affiliated First Hospital, Ningbo, Zhejiang Province, People's Republic of China.
3. Ningbo University School of Medicine, Ningbo, Zhejiang Province, People's Republic of China.
4. State Key Laboratory of Agricultural Microbiology, College of Veterinary Medicine, Huazhong Agricultural University, Wuhan, Hubei Province, People's Republic of China.
† Yuchao Zhu, Shuai Fang, Bolin Fan and Kaiwei Xu are contributed equally.
Abstract
Background: Pancreatic ductal adenocarcinoma (PDAC) is an insidious, rapidly progressing malignancy of the gastrointestinal tract. Due to its dense fibrous stroma and complex tumor microenvironment, neither of which is sensitive to radiotherapy, pancreatic adenocarcinoma is one of the malignancies with the poorest prognosis. Therefore, detailed elucidation of the inhibitory microenvironment of PDAC is essential for the development of novel therapeutic strategies.
Methods: We analyzed the association between cancer-associated fibroblasts (CAFs) and resistance to ferroptosis in PDAC using conditioned CAF medium and co-culture of pancreatic cancer cells. Abnormal cysteine metabolism was observed in CAFs using non-targeted metabolomics analysis with liquid chromatography-tandem mass spectrometry (LC-MS/MS). The regulatory effects of cysteine were investigated in PDAC cells through measurement of cell cloning, cell death, cell function, and EdU assays. The effects of exogenous cysteine intake were examined in a mouse xenograft model and the effects of the cysteine pathway on ferroptosis in PDAC were investigated by western blotting, measurement of glutathione and reactive oxygen species levels, among others.
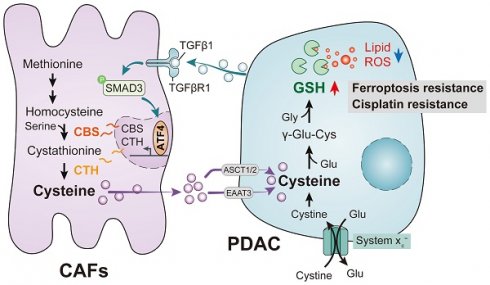
Results: It was found that CAFs played a critical role in PDAC metabolism by secreting cysteine, which could increase tumor resistance to ferroptosis. A previously unrecognized function of the sulfur transfer pathway in CAFs was identified, which increased the extracellular supply of cysteine to support glutathione synthesis and thus inducing ferroptosis resistance. Cysteine secretion by CAFs was found to be mediated by the TGF-β/SMAD3/ATF4 signaling axis.
Conclusion: Taken together, the findings demonstrate a novel metabolic relationship between CAFs and cancer cells, in which cysteine generated by CAFs acts as a substrate in the prevention of oxidative damage in PDAC and thus suggests new therapeutic targets for PDAC.
Keywords: CAF, cysteine, pancreatic cancer, metabolism, TME
 Global reach, higher impact
Global reach, higher impact