Impact Factor
Theranostics 2023; 13(1):267-277. doi:10.7150/thno.78884 This issue Cite
Research Paper
Zwitterionic rhodamine-CPT prodrug nanoparticles with GSH/H2O2 responsiveness for cancer theranostics
1. Key Lab of Biobased Polymer Materials, Shandong Provincial Education Department, College of Polymer Science and Engineering, Qingdao University of Science and Technology, Qingdao, 266042, P.R. China.
2. Chongqing Key Laboratory of Extraordinary Bond Engineering and Advanced Materials Technology, College of Materials Science and Engineering, Yangtze Normal University, Chongqing, 408100, P.R. China.
#Equal contributions to this work.
Abstract
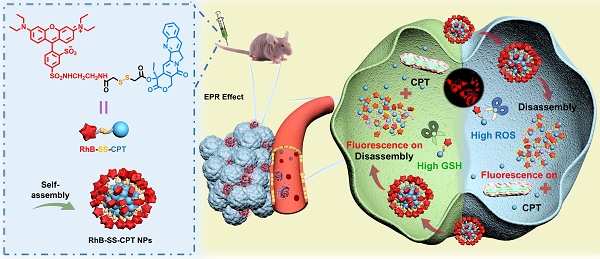
Rationale: Fluorescently traceable prodrugs, which can monitor their biodistribution in vivo and track the kinetics of drug delivery in living cells, are promising for constructing theranostic medicines. However, due to their charge and hydrophobicity, most of the fluorescently traceable prodrugs exhibit high protein binding and non-specific tissue retention affecting in vivo distribution and toxicity, with high background signals.
Methods: Herein, the zwitterionic rhodamine (RhB) and camptothecin (CPT) were bridged with a disulfide bond to construct a tumorous heterogeneity-activatable prodrug (RhB-SS-CPT). The interaction of zwitterionic RhB-SS-CPT with proteins was detected by UV and fluorescence spectroscopy, and further demonstrated by molecular docking studies. Then, intracellular tracking and cytotoxicity of RhB-SS-CPT were determined in tumor and normal cells. Finally, the in vivo biodistribution, pharmacokinetics, and anticancer efficacy of RhB-SS-CPT were evaluated in a mouse animal model.
Results: The tumorous heterogeneity-activatable RhB-SS-CPT prodrug can self-assemble into stable nanoparticles in water based on its amphiphilic structure. Particularly, the zwitterionic prodrug nanoparticles reduce the non-specific binding to generate a low background signal for better identification of cancerous lesions, achieve rapid internalization into cancer cells, selectively release bioactive CPT as a cytotoxic agent in response to high levels of GSH and H2O2, and exhibit high fluorescence that contributes to the visual chemotherapy modality. In addition, the RhB-SS-CPT prodrug nanoparticles show longer circulation time and better antitumor activity than free CPT in vivo. Interestingly, the zwitterionic nature allows RhB-SS-CPT to be excreted through the renal route, with fewer side effects.
Conclusions: Zwitterionic features and responsive linkers are important considerations for constructing potent prodrugs, which provide some useful insights to design the next-generation of theranostic prodrugs for cancer.
Keywords: Camptothecin prodrug, zwitterion, disulfide bond, self-assembly, nanomedicine
 Global reach, higher impact
Global reach, higher impact