Impact Factor
Theranostics 2022; 12(17):7237-7249. doi:10.7150/thno.77345 This issue Cite
Research Paper
MMP 9-instructed assembly of bFGF nanofibers in ischemic myocardium to promote heart repair
1. Department of Anesthesiology, The Second Affiliated Hospital of Anhui Medical University, 678 Furong Road, Hefei 230601, PR China.
2. Key Laboratory of Anesthesiology and Perioperative Medicine of Anhui Higher Education Institutes, Anhui Medical University, 678 Furong Road, Hefei 230601, PR China.
3. Inflammation and Immune Mediated Diseases Laboratory of Anhui Province, Anhui Medical University, 81 Meishan Road, Hefei 230032, PR China.
4. State Key Laboratory of Bioelectronics School of Biological Sciences and Medical Engineering Southeast University, 2 Sipailou Road, Nanjing 210096, PR China.
* These three authors contributed equally to this work.
Abstract
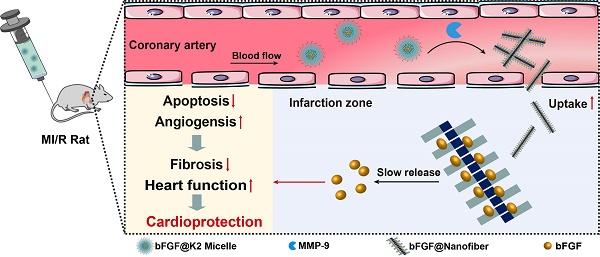
Background: The only effective treatment for myocardial infarction (MI) is the timely restoration of coronary blood flow in the infarcted area, but further reperfusion exacerbates myocardial injury and leads to distal coronary no-reflow, which affects patient prognosis. Angiogenesis could be an important therapeutic strategy for re-establishing the blood supply to save the ischemic myocardium after MI. Basic fibroblast growth factor (bFGF) has been shown to promote angiogenesis. However, direct intravenous administration of bFGF is not a viable option given its poor half-life in vivo.
Methods: Herein, we developed a peptide Lys-Lys-Pro-Leu-Gly-Leu-Ala-Gly-Phe-Phe (K2) to encapsulate bFGF to form bFGF@K2 micelle and proposed an enzyme-instructed self-assembly (EISA) strategy to deliver and slowly release bFGF in the ischemic myocardium.
Results: The bFGF@K2 micelle exerted a stronger cardioprotective effect than free bFGF in a rat model of myocardial ischemia-reperfusion (MI/R). In vitro results revealed that the bFGF@K2 micelle could be cleaved by matrix metallopeptidase 9 (MMP-9) to yield bFGF@Nanofiber through amphipathic changes. In vivo experiments indicated that intravenous administration of bFGF@K2 micelle could lead to their restructuring into bFGF@Nanofiber and long term retention of bFGF in the ischemic myocardium of rat due to high expression of MMP-9 and assembly-induced retention (AIR) effect, respectively. Twenty-eight days after MI/R model establishment, bFGF@K2 micelle treatment significantly reduced fibrosis and improved cardiac function of the rats.
Conclusion: We predict that our strategy could be applied in clinic for MI treatment in the future.
Keywords: Assembly, Basic fibroblast growth factor, Matrix metallopeptidase 9, Myocardial ischemia-reperfusion, Slow-release
 Global reach, higher impact
Global reach, higher impact