13.3
Impact Factor
Theranostics 2022; 12(14):6422-6436. doi:10.7150/thno.77350 This issue Cite
Research Paper
Circular RNA cancer vaccines drive immunity in hard-to-treat malignancies
1. Institute for Immunology and School of Medicine, Tsinghua University, Beijing 100084, China.
2. Key Laboratory of Bioorganic Phosphorus Chemistry & Chemical Biology, Department of Chemistry, Tsinghua University, Beijing, 100084, China.
3. School of Pharmaceutical Sciences, Tsinghua University, Beijing 100084, China.
4. Tsinghua-Peking Center for Life Sciences, Beijing 100084, China.
*These authors contributed equally to the manuscript.
Abstract
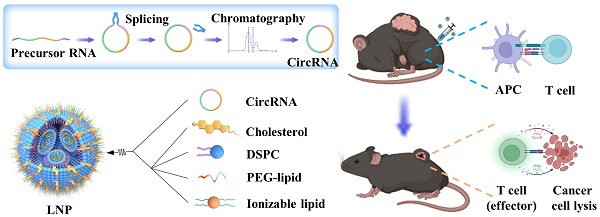
Rationale: Messenger RNA (mRNA) vaccine outperforms other kinds of cancer immunotherapy due to its high response rates, easy preparation, and wide applicability, which is considered as one of the most promising forms of next-generation cancer therapies. However, the inherent instability and insufficient protein expression duration of mRNA limit the efficacy and widespread application of the vaccine.
Methods: Here, we first tested the possibility of a novel circular RNA (circRNA) platform for protein expression and compare its duration with linear RNA. Then, we developed a lipid nanoparticle (LNP) system for circRNA delivery in vitro and in vivo. Next, the innate and adaptive immune response of circRNA-LNP complex was evaluated in vivo. The anti-tumor efficacy of circRNA-LNP was further confirmed in three tumor models. Finally, the possibility of combination therapy with circRNA-LNP and adoptive cell transfer therapy was further investigated in a late-stage tumor model.
Results: We successfully increased the stability of the RNA vaccine by circularizing the linear RNA molecules to form highly stable circRNA molecules which exhibited durable protein expression ability. By encapsulating the antigen-coding circRNA in LNP enabling in vivo expression, we established a novel circRNA vaccine platform, which was capable of triggering robust innate and adaptive immune activation and showed superior anti-tumor efficacy in multiple mouse tumor models.
Conclusions: Overall, our circRNA vaccine platform provides a novel prospect for the development of cancer RNA vaccines in a wide range of hard-to-treat malignancies.
Keywords: circular RNA, cancer vaccines, lipid nanoparticles, hard-to-treat malignancies, tumor immunotherapy
 Global reach, higher impact
Global reach, higher impact