Impact Factor
Theranostics 2022; 12(12):5574-5595. doi:10.7150/thno.70754 This issue Cite
Research Paper
Microbial metabolite restricts 5-fluorouracil-resistant colonic tumor progression by sensitizing drug transporters via regulation of FOXO3-FOXM1 axis
1. Department of Microbiology and Immunology, Brown Cancer Center, Center for Microbiomics, Inflammation and Pathogenicity, University of Louisville, Louisville, KY, USA.
2. Department of Radiology, Center for Predictive Medicine, University of Louisville, Louisville, KY, USA.
3. Institute for Stem Cell Biology and Regenerative Medicine (inStem), GKVK campus, Bangalore, Karnataka 560065, India.
4. Department of Molecular Diagnostics and Experimental Therapeutics, City of Hope Comprehensive Cancer Center, Duarte, CA, USA.
Abstract
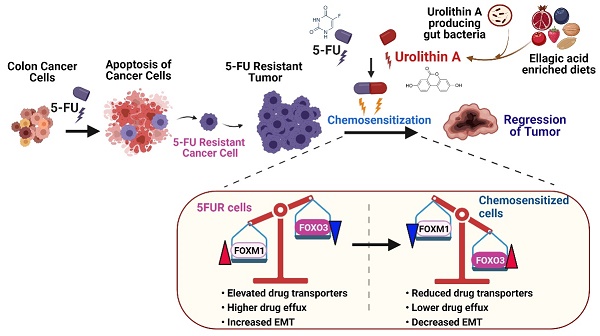
The survival rate of colorectal cancer patients is adversely affected by the selection of tumors resistant to conventional anti-cancer drugs such as 5-fluorouracil (5FU). Although there is mounting evidence that commensal gut microbiota is essential for effective colon cancer treatment, the detailed molecular mechanisms and the role of gut microbial metabolites remain elusive. The goal of this study is to decipher the impact and mechanisms of gut microbial metabolite, urolithin A (UroA) and its structural analogue, UAS03 on reversal of 5FU-resistant (5FUR) colon cancers.
Methods: We have utilized the SW480 and HCT-116 parental (5FU-sensitive) and 5FUR colon cancer cells to examine the chemosensitization effects of UroA or UAS03 by using both in vitro and in vivo models. The effects of mono (UroA/UAS03/5FU) and combinatorial therapy (UroA/UAS03 + 5FU) on cell proliferation, apoptosis, cell migration and invasion, regulation of epithelial mesenchymal transition (EMT) mediators, expression and activities of drug transporters, and their regulatory transcription factors were examined using molecular, cellular, immunological and flowcytometric methods. Further, the anti-tumor effects of mono/combination therapy (UroA or UAS03 or 5FU or UroA/UAS03 + 5FU) were examined using pre-clinical models of 5FUR-tumor xenografts in NRGS mice and azoxymethane (AOM)-dextran sodium sulfate (DSS)-induced colon tumors.
Results: Our data showed that UroA or UAS03 in combination with 5FU significantly inhibited cell viability, proliferation, invasiveness as well as induced apoptosis of the 5FUR colon cancer cells compared to mono treatments. Mechanistically, UroA or UAS03 chemosensitized the 5FUR cancer cells by downregulating the expression and activities of drug transporters (MDR1, BCRP, MRP2 and MRP7) leading to a decrease in the efflux of 5FU. Further, our data suggested the UroA or UAS03 chemosensitized 5FUR cancer cells to 5FU treatment through regulating FOXO3-FOXM1 axis. Oral treatment with UroA or UAS03 in combination with low dose i.p. 5FU significantly reduced the growth of 5FUR-tumor xenografts in NRGS mice. Further, combination therapy significantly abrogated colonic tumors in AOM-DSS-induced colon tumors in mice.
Conclusions: In summary, gut microbial metabolite UroA and its structural analogue UAS03 chemosensitized the 5FUR colon cancers for effective 5FU chemotherapy. This study provided the novel characteristics of gut microbial metabolites to have significant translational implications in drug-resistant cancer therapeutics.
Keywords: Chemoresistant colon cancer, 5-Fluorouracil, Microbial metabolite, Urolithin A, Chemosensitization, drug transporters, FOXO3-FOXM1 axis
 Global reach, higher impact
Global reach, higher impact