Impact Factor
Theranostics 2022; 12(12):5451-5469. doi:10.7150/thno.72248 This issue Cite
Research Paper
Targeting the splicing factor NONO inhibits GBM progression through GPX1 intron retention
1. Department of Neurosurgery, Qilu Hospital, Cheeloo College of Medicine and Institute of Brain and Brain-Inspired Science, Shandong University, Jinan, 250012, China
2. Jinan Microecological Biomedicine Shandong Laboratory, Jinan, 250117, China and Shandong Key Laboratory of Brain Function Remodeling, Jinan, 250012, China
3. Medical Integration and Practice Center, Cheeloo College of Medicine, Shandong University, Jinan, 250012, China
4. University of Pittsburgh Medical Center, Hillman Cancer Center, Pittsburgh, PA, USA
5. Department of Biomedicine, University of Bergen, Jonas Lies vei 91, 5009 Bergen, Norway
*These authors contributed equally to this work as senior authors.
Abstract
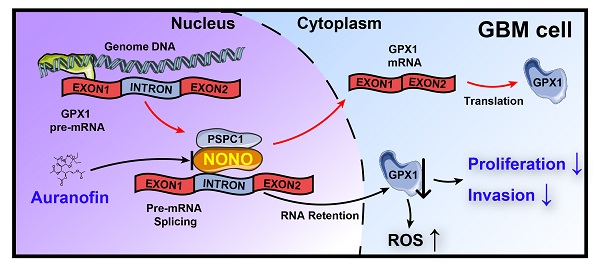
Background: Splicing factors are essential for nascent pre-mRNA processing and critical in cancer progression, suggesting that proteins with splicing functions represent potential molecular targets for cancer therapy. Here, we investigate the role of splicing factors in glioblastoma multiforme (GBM) progression and the possibility of targeting them for the treatment of the disease.
Methods: The TCGA and CGGA public databases were used to screen for differentially expressed mRNA splicing factors. Immunohistochemistry and qRT-PCR were used to analyze the expression of non-POU domain-containing octamer-binding protein (NONO), a Drosophila behavior human splicing (DBHS) protein. Knockdown/overexpression of NONO with siRNA and lentiviral expression constructs was used to examine cell growth, apoptosis, and invasion in GBM cells. RNA sequencing was used to identify potential downstream molecular targets of NONO. RIP-PCR and RNA pulldown were used to determine the interaction between NONO and pre-mRNA. JC-1 staining and the seahorse assay were performed to assess redox homeostasis.
Results: Expression of NONO was increased in GBM samples and associated with poor survival in patients (P = 0.04). Knockdown of NONO suppressed GBM growth, and overexpression of NONO promoted GBM tumorigenesis in vitro and in vivo. RNA sequencing-based transcriptomic profiling confirmed that knockdown of NONO in U251 and P3 cells resulted in global intron retention of pre-mRNA and led to abnormal splicing of specific pre-mRNAs for GPX1 and CCN1. NONO bound to a consensus motif in the intron of GPX1 pre-mRNA in association with another DBHS protein family member, PSPC1. Knockdown of NONO impaired tumor growth, invasion, and redox homeostasis through aberrant splicing of GPX1. Finally, Auranofin, a small molecule inhibitor of NONO, suppressed GBM tumor growth in an orthotopic xenograft model in mice.
Conclusions: We demonstrated that intron retention was a critical alternative RNA splicing event to occur in GBM progression, and that NONO was a key regulator of mRNA splicing in GBM. Targeting NONO represents a novel, potential therapeutic strategy for GBM treatment.
Keywords: Glioblastoma multiforme, NONO, mRNA splicing, GPX1, Auranofin
 Global reach, higher impact
Global reach, higher impact