13.3
Impact Factor
Theranostics 2022; 12(12):5389-5403. doi:10.7150/thno.72614 This issue Cite
Research Paper
Subpial delivery of adeno-associated virus 9-synapsin-caveolin-1 (AAV9-SynCav1) preserves motor neuron and neuromuscular junction morphology, motor function, delays disease onset, and extends survival in hSOD1G93A mice
1. Department of Anesthesiology, VA San Diego Healthcare System, San Diego, CA, USA
2. Department of Anesthesiology, University of California San Diego, La Jolla, CA, USA
3. Neuroregeneration Laboratory, Department of Anesthesiology, University of California San Diego, La Jolla, CA, USA
4. Department of Anesthesiology, University of the Ryukyus, Okinawa, Japan
5. Department of Anesthesiology, Sapporo Medical University School of Medicine, Sapporo, Japan
6. Department of Anesthesiology and Pain Medicine, Yonsei University College of Medicine, Seoul, South Korea
7. Department of Anesthesiology, Nagasaki University Hospital, Nagasaki, Japan
* Shanshan Wang and Taiga Ichinomaya share co-1st authorship.
Abstract
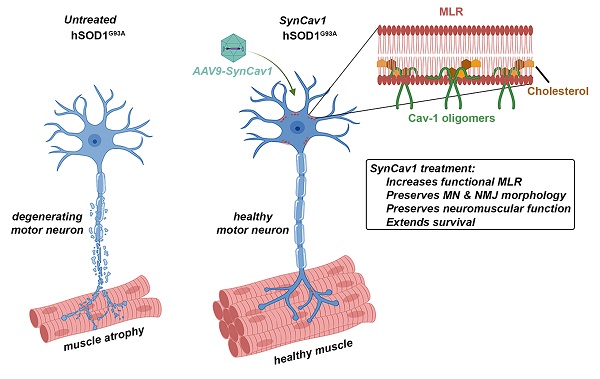
Elevating neuroprotective proteins using adeno-associated virus (AAV)-mediated gene delivery shows great promise in combating devastating neurodegenerative diseases. Amyotrophic lateral sclerosis (ALS) is one such disease resulting from loss of upper and lower motor neurons (MNs) with 90-95% of cases sporadic (SALS) in nature. Due to the unknown etiology of SALS, interventions that afford neuronal protection and preservation are urgently needed. Caveolin-1 (Cav-1), a membrane/lipid rafts (MLRs) scaffolding and neuroprotective protein, and MLR-associated signaling components are decreased in degenerating neurons in postmortem human brains. We previously showed that, when crossing our SynCav1 transgenic mouse (TG) with the mutant human superoxide dismutase 1 (hSOD1G93A) mouse model of ALS, the double transgenic mouse (SynCav1 TG/hSOD1G93A) exhibited better motor function and longer survival. The objective of the current study was to test whether neuron-targeted Cav-1 upregulation in the spinal cord using AAV9-SynCav1 could improve motor function and extend longevity in mutant humanized mouse and rat (hSOD1G93A) models of familial (F)ALS.
Methods: Motor function was assessed by voluntary running wheel (RW) in mice and forelimb grip strength (GS) and motor evoked potentials (MEP) in rats. Immunofluorescence (IF) microscopy for choline acetyltransferase (ChAT) was used to assess MN morphology. Neuromuscular junctions (NMJs) were measured by bungarotoxin-a (Btx-a) and synaptophysin IF. Body weight (BW) was measured weekly, and the survival curve was determined by Kaplan-Meier analysis.
Results: Following subpial gene delivery to the lumbar spinal cord, male and female hSOD1G93A mice treated with SynCav1 exhibited delayed disease onset, greater running-wheel performance, preserved spinal alpha-motor neuron morphology and NMJ integrity, and 10% increased longevity, independent of affecting expression of the mutant hSOD1G93A protein. Cervical subpial SynCav1 delivery to hSOD1G93A rats preserved forelimb GS and MEPs in the brachial and gastrocnemius muscles.
Conclusion: In summary, subpial delivery of SynCav1 protects and preserves spinal motor neurons, and extends longevity in a familial mouse model of ALS without reducing the toxic monogenic component. Furthermore, subpial SynCav1 delivery preserved neuromuscular function in a rat model of FALS. The latter findings strongly indicate the therapeutic applicability of SynCav1 to treat ALS attributed to monogenic (FALS) and potentially in sporadic cases (i.e., SALS).
Keywords: caveolin-1, membrane/lipid raft (MLRs), gene therapy, hSOD1G93A, amyotrophic lateral sclerosis, motor neuron, neuromuscular junction
 Global reach, higher impact
Global reach, higher impact