Impact Factor
Theranostics 2022; 12(12):5337-5349. doi:10.7150/thno.75100 This issue Cite
Research Paper
XBP1 is required in Th2 polarization induction in airway allergy
1. Department of Otolaryngology. Longgang E.N.T Hospital & Shenzhen Key Laboratory of E.N.T, Institute of E.N.T Shenzhen, China.
2. Guangdong Provincial Key Laboratory of Regional Immunity and Diseases, Shenzhen, China.
3. Institute of Allergy & Immunology, Shenzhen University School of Medicine, State Key Laboratory of Respiratory Disease Allergy Division at Shenzhen University, Shenzhen, China.
4. Department of Respirology and Allergy, Third Affiliated Hospital of Shenzhen University, Shenzhen, China.
#Equally contributed to this work.
Abstract
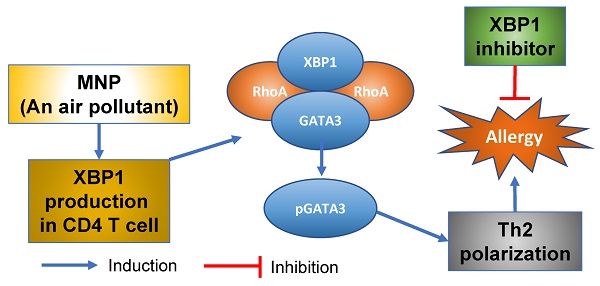
Rationale: Th2 polarization plays a central role in the pathogenesis of allergic diseases such as airway allergy. The underlying mechanism is not fully understood yet. X-box-binding protein-1 (XBP1) can regulate immune cell activities upon exposing stressful events. The role of XBP1 in the development of Th2 polarization has not yet been explored.
Methods: Mice carrying Xbp1-deficient CD4+ T cells were employed to observe the role of XBP1 in the induction of airway allergy. A cell culture model was established to evaluate the role of XBP1 in facilitating the Th2 lineage commitment.
Results: We found that Xbp1 ablation in CD4+ T cells prevented induction of Th2 polarization in the mouse airway tract. XBP1 was indispensable in the Th2 lineage commitment. XBP1 mediated the effects of 3-methyl-4-nitrophenol (MNP) on facilitating inducing antigen-specific Th2 response in the airways. Exposure to MNP induced expression of XBP1 in CD4+ T cells. RhoA facilitated the binding between XBP1 and GATA3 in CD4+ T cells. XBP1 induced GATA3 phosphorylation to promote the Il4 gene transcription. Modulation of the RhoA/XBP1 axis mitigated experimental allergic response in the mouse airways.
Conclusions: A potential therapeutic target, XBP1, was identified in this study. XBP1 was required in the development of skewed Th2 response in the airways. Inhibiting XBP1 alleviated Th2 polarization-related immune inflammation in the airways. The data suggest that inhibiting XBP1 has the translation potential for the treatment of airway allergy.
Keywords: Airway allergy, air pollution, CD4+ T cells, Th2 polarization, XBP1.
 Global reach, higher impact
Global reach, higher impact