Impact Factor
Theranostics 2022; 12(12):5258-5271. doi:10.7150/thno.72037 This issue Cite
Research Paper
DCLK1 promotes colorectal cancer stemness and aggressiveness via the XRCC5/COX2 axis
1. School of Life Sciences, Gwangju Institute of Science and Technology, Gwangju, 61005, Republic of Korea.
2. Cell Logistics Research Center, Gwangju Institute of Science and Technology, Gwangju, 61005, Republic of Korea.
3. Korea Chemical Bank, Korea Research Institute of Chemical Technology, Daejeon, 34114, Republic of Korea.
4. Therapeutics and Biotechnology Division, Korea Research Institute of Chemical Technology, Daejeon, 34114, Republic of Korea.
5. Department of Hemato-Oncology, Chonnam National University Medical School, Gwangju, 61469, Republic of Korea.
6. Department of Pathology, Chonnam National University Medical School, Gwangju, 61469, Republic of Korea.
#Current address: Institute for Basic Science, Center for Genome Engineering, 55, Expo-ro, Yuseong-gu, Daejeon, Korea, 34126.
*These authors contributed equally to this work.
Abstract
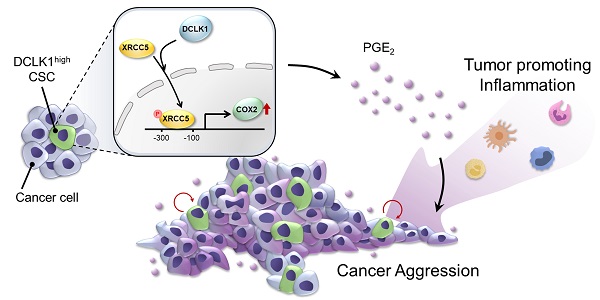
Rationale: Doublecortin-like kinase 1 (DCLK1) is a serine/threonine kinase that selectively marks cancer stem-like cells (CSCs) and promotes malignant progression in colorectal cancer (CRC). However, the exact molecular mechanism by which DCLK1 drives the aggressive phenotype of cancer cells is incompletely determined.
Methods: Here, we performed comprehensive genomics and proteomics analyses to identify binding proteins of DCLK1 and discovered X-ray repair cross-complementing 5 (XRCC5). Thus, we explored the biological role and downstream events of the DCLK1/XRCC5 axis in human CRC cells and CRC mouse models.
Results: The results of comprehensive bioinformatics analyses suggested that DCLK1-driven CRC aggressiveness is linked to inflammation. Mechanistically, DCLK1 bound and phosphorylated XRCC5, which in turn transcriptionally activated cyclooxygenase-2 expression and enhanced prostaglandin E2 production; these events collectively generated the inflammatory tumor microenvironment and enhanced the aggressive behavior of CRC cells. Consistent with the discovered mechanism, inhibition of DCLK1 kinase activity strongly impaired the tumor seeding and growth capabilities in CRC mouse models.
Conclusion: Our study illuminates a novel mechanism that mediates the pro-inflammatory function of CSCs in driving the aggressive phenotype of CRC, broadening the biological function of DCLK1 in CRC.
Keywords: Cancer stem cells, Inflammatory tumor microenvironment, Doublecortin-like kinase 1, Prostaglandin E2
 Global reach, higher impact
Global reach, higher impact