Impact Factor
Theranostics 2022; 12(10):4802-4817. doi:10.7150/thno.73746 This issue Cite
Research Paper
N6-methyladenosine-mediated LDHA induction potentiates chemoresistance of colorectal cancer cells through metabolic reprogramming
1. The Second Affiliated Hospital of Chengdu Medical College, China National Nuclear Corporation 416 Hospital, Chengdu 610051, China.
2. School of Biological Sciences and Technology, Chengdu Medical College, Chengdu 610500, China.
3. Sun Yat-sen University Cancer Center; State Key Laboratory of Oncology in South China; Collaborative Innovation Center for Cancer Medicine, Guangzhou 510060, China.
4. School of Pharmacy, Chengdu Medical College, Chengdu 610500, China.
#These authors contributed equally to this work.
Abstract
Background: Chemoresistance to 5-fluorouracil (5-FU) is a major barrier to influence the treatment efficiency of colorectal cancer (CRC) patients, while the precise molecular mechanisms underlying 5-FU resistance remain to be fully elucidated.
Methods: The metabolic profiles including ATP generation, glucose consumption, lactate generation, and oxygen consumption rate (OCR) in 5-FU resistant CRC cells were compared with those in their parental cells. Subsequently, a series of in vitro and in vivo experiments were carried out to investigate the mechanisms responsible for metabolic reprogramming of 5-FU resistant CRC cells.
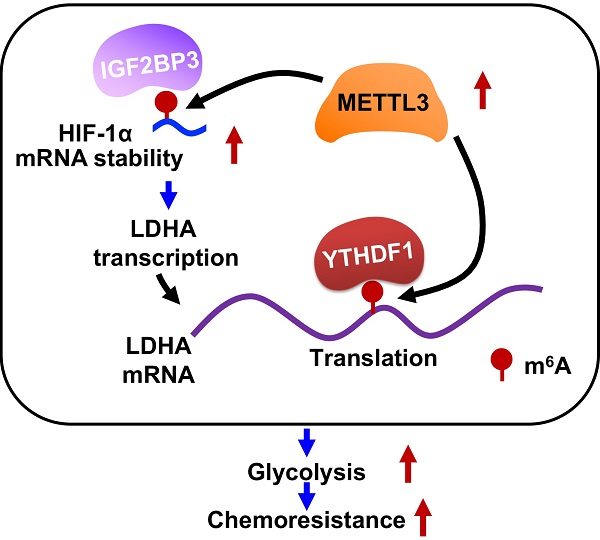
Results: We found that 5-FU resistant CRC cells showed increased levels of ATP generation, glucose consumption, lactate generation, and OCR as compared with those in their parental cells. Further, increased levels of mRNA N6-methyladenosine (m6A) and methyltransferase-like 3 (METTL3) were observed in 5-FU resistant CRC cells. Inhibition or knockdown of METTL3 can suppress glycolysis and restore chemosensitivity of 5-FU resistant CRC cells. Mechanistically, METTL3 enhances the expression of LDHA, which catalyzes the conversion of pyruvate to lactate, to trigger glycolysis and 5-FU resistance. METTL3 can increase the transcription of LDHA via stabilizing mRNA of hypoxia-inducible factor (HIF-1α), further, METTL3 also triggers the translation of LDHA mRNA via methylation of its CDS region and recruitment of YTH domain-containing family protein 1 (YTHDF1). Targeted inhibition of METTL3/LDHA axis can significantly increase the in vitro and in vivo 5-FU sensitivity of CRC cells.
Conclusion: Our study indicates that METTL3/LDHA axis-induced glucose metabolism is a potential therapy target to overcome 5-FU resistance in CRC cells.
Keywords: Glycolysis, METTL3, 5-FU, LDHA, Chemoresistance
 Global reach, higher impact
Global reach, higher impact