13.3
Impact Factor
Theranostics 2022; 12(10):4753-4766. doi:10.7150/thno.72456 This issue Cite
Research Paper
MiR-155 deficiency protects renal tubular epithelial cells from telomeric and genomic DNA damage in cisplatin-induced acute kidney injury
1. Institute of Nephrology, Zhong Da Hospital, Southeast University School of Medicine, Nanjing, Jiangsu, China.
2. Department of Pediatric Nephrology, the Second Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu, China.
3. Nanjing Medical University, Nanjing, Jiangsu, China.
4. Institute of Nephrology, Taizhou Clinical Medical School of Nanjing Medical University (Taizhou People's Hospital), Taizhou, Jiangsu, China.
5. Department of Pediatric Nephrology, Affiliated Maternity and Child Health Care Hospital of Nantong University, Nantong, Jiangsu, China.
Abstract
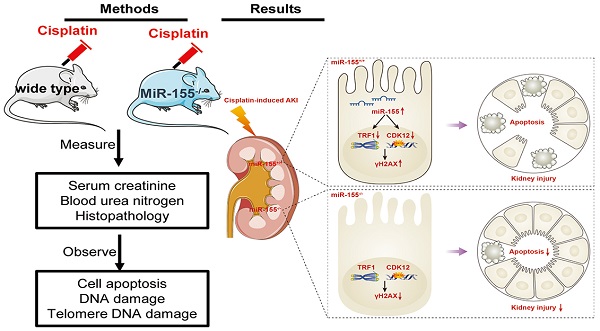
Rationale: Cisplatin nephrotoxicity is an important cause of acute kidney injury (AKI), limiting cisplatin application in cancer therapy. Growing evidence has suggested that genome instability, telomeric dysfunction, and DNA damage were involved in the tubular epithelial cells (TECs) damage in cisplatin-induced AKI (cAKI). However, the exact mechanism is largely unknown.
Methods: We subjected miR-155-/- mice and wild-type controls, as well as HK-2 cells, to cAKI models. We assessed kidney function and injury with standard techniques. The cell apoptosis and DNA damage of TECs were evaluated both in vivo and in vitro. Telomeres were measured by the fluorescence in situ hybridization.
Results: The expression level of miR-155 was upregulated in cAKI. Inhibition of miR-155 expression protected cisplatin-induced AKI both in vivo and in vitro. Compared with wild-type mice, miR-155-/- mice had reduced mortality, improved renal function and pathological damage after cisplatin intervention. Moreover, inhibition of miR-155 expression attenuated TECs apoptosis and DNA damage. These protective effects were caused by increasing expression of telomeric repeat binding factor 1 (TRF1) and cyclin-dependent kinase 12 (CDK12), thereby limiting the telomeric dysfunction and the genomic DNA damage in cAKI.
Conclusion: We demonstrated that miR-155 deficiency could significantly attenuate pathological damage and mortality in cAKI through inhibition of TECs apoptosis, genome instability, and telomeric dysfunction, which is possibly regulated by the increasing expression of TRF1 and CDK12. This study will provide a new molecular strategy for the prevention of cAKI.
Keywords: MiR-155, DNA damage, AKI, TRF1, CDK12
 Global reach, higher impact
Global reach, higher impact