13.3
Impact Factor
Theranostics 2022; 12(10):4498-4512. doi:10.7150/thno.72818 This issue Cite
Research Paper
Specific (sialyl-)Lewis core 2 O-glycans differentiate colorectal cancer from healthy colon epithelium
1. Leiden University Medical Center, Center for Proteomics and Metabolomics, Postbus 9600, 2300 RC Leiden, The Netherlands
2. Leiden University Medical Center, Department of Surgery, Postbus 9600, 2300 RC Leiden, The Netherlands
3. Leiden University Medical Center, Department of Pathology, Postbus 9600, 2300 RC Leiden, The Netherlands
4. Department of Chemical Biology and Drug Discovery, Utrecht University, Universiteitsweg 99, 3584 CG Utrecht, The Netherlands
Abstract
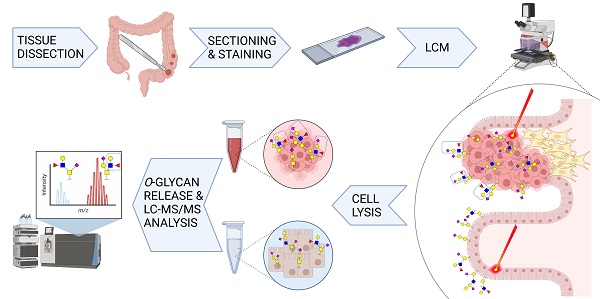
Cells are covered with a dense layer of carbohydrates, some of which are solely present on neoplastic cells. The so-called tumor-associated carbohydrate antigens (TACAs) are increasingly recognized as promising targets for immunotherapy. These carbohydrates differ from those of the surrounding non-cancerous tissues and contribute to the malignant phenotype of the cancer cells by promoting proliferation, metastasis, and immunosuppression. However, due to tumor tissue heterogeneity and technological limitations, TACAs are insufficiently explored.
Methods: A workflow was established to decode the colorectal cancer (CRC)-associated O-linked glycans from approximately 20,000 cell extracts. Extracts were obtained through laser capture microdissection of formalin fixed paraffin embedded tissues of both primary tumors and metastatic sites, and compared to healthy colon mucosa from the same patients. The released O-glycans were analyzed by porous graphitized carbon liquid chromatography-tandem mass spectrometry in negative ion mode.
Results: Distinctive O-glycosylation features were found in cancerous, stromal and normal colon mucosal regions. Over 100 O-linked glycans were detected in cancerous regions with absence in normal mucosa. From those, six core 2 O-glycans were exclusively found in more than 33% of the cancers, carrying the terminal (sialyl-)LewisX/A antigen. Moreover, two O-glycans were present in 72% of the analyzed cancers and 94% of the investigated cancers expressed at least one of these two O-glycans. In contrast, normal colon mucosa predominantly expressed core 3 O-glycans, carrying α2-6-linked sialylation, (sulfo-)LewisX/A and Sda antigens.
Conclusion: In this study, we present a novel panel of highly specific TACAs, based upon differences in the glycomic profiles between CRC and healthy colon mucosa. These TACAs are promising new targets for development of innovative cancer immune target therapies and lay the foundation for the targeted treatment of CRC.
Keywords: Tumor associated carbohydrate antigens (TACAs), mass spectrometry, glycomics, therapeutic target
 Global reach, higher impact
Global reach, higher impact