Impact Factor
Theranostics 2022; 12(9):4446-4458. doi:10.7150/thno.63205 This issue Cite
Research Paper
Molecular Imaging-Derived Biomarker of Cardiac Nerve Integrity — Introducing High NET Affinity PET Probe 18F-AF78
1. Nuclear Medicine, Faculty of Medicine, University of Augsburg, Augsburg, Germany.
2. Department of Nuclear Medicine and Comprehensive Heart Failure Center, University Hospital of Würzburg, Würzburg, Germany.
3. Division of Nuclear Medicine, The Russell H Morgan Department of Radiology and Radiological Science, Johns Hopkins University School of Medicine, Baltimore, MD, U.S.
4. Department of Systems and Informatics, Hokkaido Information University, Ebetsu, Hokkaido, Japan.
5. Faculty of Medicine, Dentistry and Pharmaceutical Sciences, Okayama University, Okayama, Japan.
6. Institute of Pharmacy and Food Chemistry, University of Würzburg, Würzburg, Germany.
*Equal contribution
Abstract
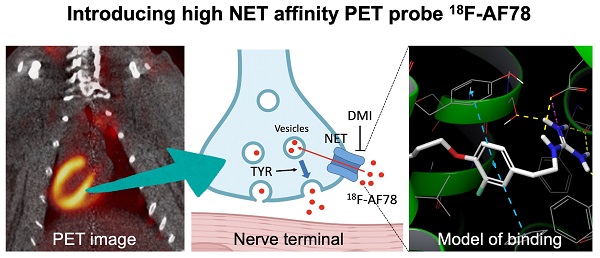
Background: Radiolabeled agents that are substrates for the norepinephrine transporter (NET) can be used to quantify cardiac sympathetic nervous conditions and have been demonstrated to identify high-risk congestive heart failure (HF) patients prone to arrhythmic events. We aimed to fully characterize the kinetic profile of the novel 18F-labeled NET probe AF78 for PET imaging of the cardiac sympathetic nervous system (SNS) among various species.
Methods: 18F-AF78 was compared to norepinephrine (NE) and established SNS radiotracers by employing in vitro cell assays, followed by an in vivo PET imaging approach with healthy rats, rabbits and nonhuman primates (NHPs). Additionally, chase protocols were performed in NHPs with NET inhibitor desipramine (DMI) and the NE releasing stimulator tyramine (TYR) to investigate retention kinetics in cardiac SNS.
Results: Relative to other SNS radiotracers, 18F-AF78 showed higher transport affinity via NET in a cell-based competitive uptake assay (IC50 0.42 ± 0.14 µM), almost identical to that of NE (IC50, 0.50 ± 0.16 µM, n.s.). In rabbits and NHPs, initial cardiac uptake was significantly reduced by NET inhibition. Furthermore, cardiac tracer retention was not affected by a DMI chase protocol but was markedly reduced by intermittent TYR chase, thereby suggesting that 18F-AF78 is stored and can be released via the synaptic vesicular turnover process. Computational modeling hypothesized the formation of a T-shaped π-π stacking at the binding site, suggesting a rationale for the high affinity of 18F-AF78.
Conclusion: 18F-AF78 demonstrated high in vitro NET affinity and advantageous in vivo radiotracer kinetics across various species, indicating that 18F-AF78 is an SNS imaging agent with strong potential to guide specific interventions in cardiovascular medicine.
Keywords: norepinephrine transporter, sympathetic nervous system, cardiac innervation imaging, radiotracer kinetics, nonhuman primates, T-shaped π-π stacking
 Global reach, higher impact
Global reach, higher impact