13.3
Impact Factor
Theranostics 2022; 12(9):4250-4268. doi:10.7150/thno.68996 This issue Cite
Research Paper
pH-responsive hybrid platelet membrane-coated nanobomb with deep tumor penetration ability and enhanced cancer thermal/chemodynamic therapy
1. MOE Key Laboratory of Macromolecular Synthesis and Functionalization Department of Polymer Science and Engineering, Zhejiang University, Hangzhou 310027, China.
2. Department of Hepatobiliary and Pancreatic Surgery, the Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou 310009, China.
3. Department of Infectious Disease, Shulan (Hangzhou) Hospital Affiliated to Zhejiang Shuren University, Shulan International Medical College, Hangzhou 310022, China.
*These authors contributed equally to this work.
Abstract
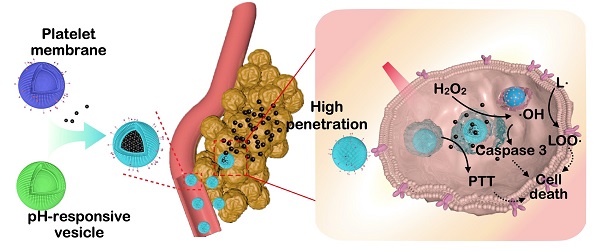
Background: Despite their outstanding properties in high surface-to-volume ratio and deep penetration, the application of ultrasmall nanoparticles for tumor theranostics remains limited because of their dissatisfied targeting performance and short blood circulation lifetime. Various synthetic materials with complex structures have been prepared as a multifunctional platform for loading ultrasmall nanoparticles. However, their use in nanomedicine is restricted because of unknown metabolic processes and potential physiological toxicity. Therefore, versatile and biocompatible nanoplatforms need to be designed through a simple yet effective method for realizing specific delivery and responsible release of ultrasmall nanoparticles.
Methods: Iron-gallic acid coordination polymer nanodots (FeCNDs) exhibits outstanding photothermal ability and Fenton catalytic performance, which can be applied for tumor inhibition via hyperthermia and reactive oxygen species. A pH-responsive platelet-based hybrid membrane (pH-HCM) was prepared via co-extrusion and acted as a safe nanoplatform to load FeCNDs (pH-HCM@FeCNDs). Subsequently, their responsive performance and penetration ability were valued considering the multicellular sphere (MCS) model in an acidic or neutral environment. Thereafter, in vivo fluorescence image was performed to assess targeting capability of pH-HCM@FeCNDs. Finally, the corresponding antitumor and antimetastatic effects on orthotropic breast cancer were investigated.
Results: In 4T1 MCS model, pH-HCM@FeCNDs group exhibited higher penetration efficiency (72.84%) than its non-responsive counterparts (17.77%) under an acidic environment. Moreover, the fluorescence intensity in pH-HCM@FeCNDs group was 3.18 times higher than that in group without targeting performance in the in vivo fluorescence image experiment. Finally, through in vivo experiments, pH-HCM@FeCNDs was confirmed to exhibit the best antitumor effect (90.33% tumor reduction) and antimetastatic effects (only 0.29% tumor coverage) on orthotropic breast cancer.
Conclusions: Hybrid cell membrane was an ideal nanoplatform to deliver nanodots because of its good responsibility, satisfactory targeting ability, and excellent biocompatibility. Consequently, this study provides novel insights into the delivery and release of nanodots in a simple but effect method.
Keywords: platelet membrane, hybrid cell membrane, nanodots, tumor penetration, combined therapy
 Global reach, higher impact
Global reach, higher impact