13.3
Impact Factor
Theranostics 2022; 12(9):4181-4199. doi:10.7150/thno.73235 This issue Cite
Research Paper
APOBEC-mediated mutagenesis is a favorable predictor of prognosis and immunotherapy for bladder cancer patients: evidence from pan-cancer analysis and multiple databases
1. Department of Oncology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, China.
2. Department of Oncology, Jiangsu Cancer Hospital, Jiangsu Institute of Cancer Research, The Affiliated Cancer Hospital of Nanjing Medical University, Nanjing, China.
3. Pancreas Center, The First Affiliated Hospital of Nanjing Medical University, Nanjing, China.
4. Department of Urology, Shanghai Ninth People's Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China.
5. Institute and Clinic for Occupational, Social and Environmental Medicine, LMU University Hospital Munich; Comprehensive Pneumology Center (CPC) Munich, German Center for Lung Research (DZL), Munich, Germany; Institute of Epidemiology, Helmholtz Zentrum München - German Research Center for Environmental Health, Neuherberg, Germany.
6. Department of Radiation Oncology, University Hospital, LMU Munich, Germany.
7. Laboratory of Chinese Herbal Pharmacology, Department of Pharmacology, Renmin Hospital; Hubei Key Laboratory of Wudang Local Chinese Medicine Research, Hubei University of Medicine, Shiyan, Hubei, China.
8. Research Unit Radiation Cytogenetics, Helmholtz Center Munich, German Research Center for Environmental Health GmbH, Neuherberg, Germany.
9. Department of Radiation Oncology, Nanfang Hospital, Southern Medical University, Guangzhou, China.
10. Departmentof Radiotherapy, Sichuan Cancer Hospital, School of Medicine, University of Electronic Science and Technology of China, Chengdu, China.
11. Department of Urology, University Hospital, LMU Munich, Germany.
12. Department of Oncology, Renmin Hospital, Hubei University of Medicine, Shiyan, Hubei, China.
13. German Cancer Consortium (DKTK), Munich, Germany.
14. Department of Pathogen Biology and Immunology, Jiangsu Province Key Laboratory of Pathogen Biology, Center for Global Health, Nanjing Medical University, Nanjing, China.
#These authors contributed equally to this work.
Abstract
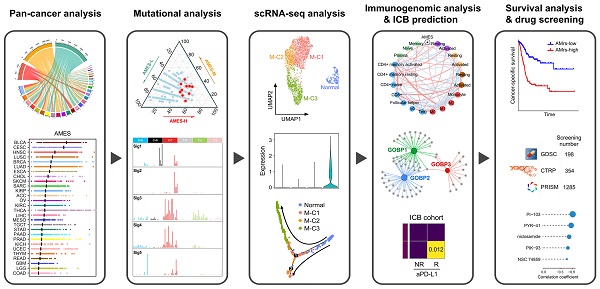
Background: The APOBEC (apolipoprotein B mRNA editing enzyme, catalytic polypeptide-like) family-mediated mutagenesis is widespread in human cancers. However, our knowledge of the biological feature and clinical relevance of APOBECs and APOBEC mutagenesis in cancers remains limited.
Methods: In this study, with a series of bioinformatic and statistical approaches, we performed a comprehensive analysis of multiple levels of data, including whole-exome sequencing (WES) and targeted next-generation sequencing (NGS), transcriptome (bulk RNA-seq and single-cell RNA-seq), immune signatures and immune checkpoint blockade (ICB) potential, patient survival and drug sensitivity, to reveal the distribution characteristics and clinical significance of APOBECs and APOBEC mutagenesis in pan-cancer especially bladder cancer (BLCA).
Results: APOBEC mutagenesis dominates in the mutational patterns of BLCA. A higher enrichment score of APOBEC mutagenesis correlates with favorable prognosis, immune activation and potential ICB response in BLCA patients. APOBEC3A and 3B play a significant role in the malignant progression and cell differentiation within the tumor microenvironment. Furthermore, using machine learning approaches, a prognostic APOBEC mutagenesis-related model was established and validated in different BLCA cohorts.
Conclusions: Our study illustrates the characterization of APOBECs and APOBEC mutagenesis in multiple cancer types and highlights its potential value as a promising biomarker for prognosis and immunotherapy in BLCA.
Keywords: APOBEC mutagenesis, Pan-cancer analysis, Immunotherapy, Prognosis
 Global reach, higher impact
Global reach, higher impact