13.3
Impact Factor
Theranostics 2022; 12(4):1570-1588. doi:10.7150/thno.67722 This issue Cite
Research Paper
Gα12 overexpression in hepatocytes by ER stress exacerbates acute liver injury via ROCK1-mediated miR-15a and ALOX12 dysregulation
1. College of Pharmacy, Seoul National University, Seoul 08826, Republic of Korea
2. Department of Clinical Pharmacology and Therapeutics, Seoul National University College of Medicine, Seoul 03080, Republic of Korea
3. Joslin Diabetes Center, Harvard Medical School, Boston, MA 02215, USA
4. Drug Information Research Institute, College of Pharmacy, Sookmyung Women's University, Seoul 04310, Republic of Korea
5. Department of Surgery, Asan Medical Center, University of Ulsan, College of Medicine, Seoul, Republic of Korea
6. College of Pharmacy and Integrated Research Institute for Drug Development, Dongguk University-Seoul, Goyang-si, Kyeonggi-do 10326, Republic of Korea
Abstract
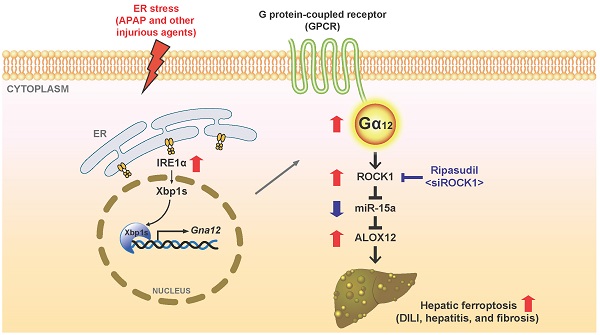
Rationale: Liver injury must be further characterized to identify novel therapeutic approaches. Endoplasmic reticulum (ER) stress may cause hepatocyte death. Gα12 affects cell viability and its expression varies depending on physiological conditions. This study investigated whether hepatocyte-specific Gα12 overexpression affects acute liver injury, and if so, what the underlying mechanisms and treatment strategies are.
Methods: All experiments were performed using human liver, hepatocytes, and toxicant injury models with Gna12 KO and/or hepatocyte-specific Gα12 overexpression. RNA-sequencing, immunoblotting, immunohistochemistry, reporter assays, and mutation assays were conducted.
Results: Hepatic Gα12 was overexpressed in mice challenged with acetaminophen or other ER stress inducers or in patients with acute liver injury or fibrosis/cirrhosis. Several Gα12 and ER-associated pathways were identified using transcriptomic analysis. Acetaminophen intoxication was characterized by lipid peroxide-induced ferroptosis and was less severe in Gα12-deficient animals and cells. Conversely, Gα12 overexpression in wild-type or Gna12 KO hepatocytes increased hepatotoxicity, promoting lipid peroxidation, inflammation, and ferroptosis. IRE1α-dependent Xbp1 transactivated Gna12. Moreover, Gα12 overexpression enhanced the ability of acetaminophen to induce ALOX12, while downregulating GPX4. The level of miR-15a, herein identified as an ALOX12 inhibitor, was decreased. siRNA knockdown or pharmacological inhibition of ROCK1 prevented dysregulation of ALOX12 and GPX4, rescuing animals from toxicant-induced ferroptosis. These changes or correlations among the targets were confirmed in human liver specimens and datasets of livers exposed to other injurious medications.
Conclusions: Gα12 overexpression by ER stress facilitates hepatocyte ferroptosis through ROCK1-mediated dysregulation of ALOX12, and miR-15a, supporting the concept that inhibition of Gα12 overexpression and/or ROCK1 axis may constitute a promising strategy for acute liver injury.
Keywords: Gα12, acetaminophen-induced liver injury, miR-15a, ALOX12, GPX4
 Global reach, higher impact
Global reach, higher impact