13.3
Impact Factor
Theranostics 2021; 11(19):9557-9570. doi:10.7150/thno.64767 This issue Cite
Research Paper
Effects of contrast-enhanced ultrasound treatment on neoadjuvant chemotherapy in breast cancer
1. Institute for Experimental Molecular Imaging, Medical Faculty, RWTH Aachen University, Aachen, Germany.
2. Chair for Medical Engineering, Department of Electrical Engineering and Information Technology, Ruhr University Bochum, Bochum, Germany.
3. Department of Obstetrics and Gynecology, Medical Faculty, RWTH Aachen University, Aachen, Germany.
4. Institute of Pathology, Medical Faculty, RWTH Aachen University, Aachen, Germany.
5. Department of Intensive Care and Intermediate Care, Medical Faculty, University Hospital RWTH Aachen, Aachen, Germany.
6. Institute for Occupational, Social and Environmental Medicine; Medical Faculty, RWTH Aachen University, Aachen, Germany.
7. Division of Imaging and Oncology, University Medical Center Utrecht, Utrecht, The Netherlands.
*These authors contributed equally to this work.
#Shared last authors.
Abstract
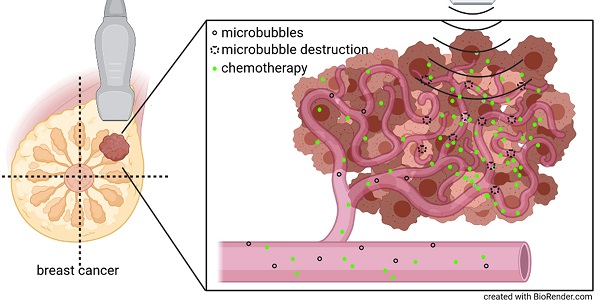
Purpose: Preclinical and clinical data indicate that contrast-enhanced ultrasound can enhance tumor perfusion and vessel permeability, thus, improving chemotherapy accumulation and therapeutic outcome. Therefore, we investigated the effects of high mechanical index (MI) contrast-enhanced Doppler ultrasound (CDUS) on tumor perfusion in breast cancer.
Methods: In this prospective study, breast cancer patients were randomly assigned to receive either 18 minutes of high MI CDUS during chemotherapy infusion (n = 6) or chemotherapy alone (n = 5). Tumor perfusion was measured before and after at least six chemotherapy cycles using motion-model ultrasound localization microscopy. Additionally, acute effects of CDUS on vessel perfusion and chemotherapy distribution were evaluated in mice bearing triple-negative breast cancer (TNBC).
Results: Morphological and functional vascular characteristics of breast cancer in patients were not significantly influenced by high MI CDUS. However, complete clinical tumor response after neoadjuvant chemotherapy was lower in high MI CDUS-treated (1/6) compared to untreated patients (4/5) and size reduction of high MI CDUS treated tumors tended to be delayed at early chemotherapy cycles. In mice with TNBC high MI CDUS decreased the perfused tumor vessel fraction (p < 0.01) without affecting carboplatin accumulation or distribution. Higher vascular immaturity and lower stromal stabilization may explain the stronger vascular response in murine than human tumors.
Conclusion: High MI CDUS had no detectable effect on breast cancer vascularization in patients. In mice, the same high MI CDUS setting did not affect chemotherapy accumulation although strong effects on the tumor vasculature were detected histologically. Thus, sonopermeabilization in human breast cancers might not be effective using high MI CDUS protocols and future applications may rather focus on low MI approaches triggering microbubble oscillations instead of destruction. Furthermore, our results show that there are profound differences in the response of mouse and human tumor vasculature to high MI CDUS, which need to be further explored and considered in clinical translation.
Keywords: CEUS, chemotherapy, breast cancer, super-resolution ultrasound, sonopermeation
 Global reach, higher impact
Global reach, higher impact