Impact Factor
Theranostics 2021; 11(19):9538-9556. doi:10.7150/thno.55954 This issue Cite
Review
Benefits of functional assays in personalized cancer medicine: more than just a proof-of-concept
1. Oncomedics SAS, 1 avenue d'ESTER, F-87069 Limoges, France.
2. Centre hospitalier universitaire Dupuytren, Service de chirurgie digestive, générale et endocrinienne, 2 avenue Martin Luther King, F-87042 Limoges, France.
3. EA3842/CAPTuR (Contrôle de l'Activation cellulaire, Progression Tumorale et Résistance thérapeutique), Faculté de Médecine, 2 rue du Docteur Marcland, F-87025 Limoges cedex, France.
Abstract
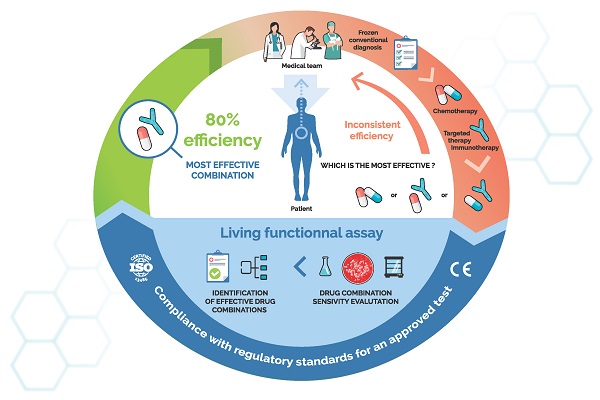
As complex and heterogeneous diseases, cancers require a more tailored therapeutic management than most pathologies. Recent advances in anticancer drug development, including the immuno-oncology revolution, have been too often plagued by unsatisfying patient response rates and survivals. In reaction to this, cancer care has fully transitioned to the “personalized medicine” concept. Numerous tools are now available tools to better adapt treatments to the profile of each patient. They encompass a large array of diagnostic assays, based on biomarkers relevant to targetable molecular pathways. As a subfamily of such so-called companion diagnostics, chemosensitivity and resistance assays represent an attractive, yet insufficiently understood, approach to individualize treatments. They rely on the assessment of a composite biomarker, the ex vivo functional response of cancer cells to drugs, to predict a patient's outcome. Systemic treatments, such as chemotherapies, as well as targeted treatments, whose efficacy cannot be fully predicted yet by other diagnostic tests, may be assessed through these means. The results can provide helpful information to assist clinicians in their decision-making process. We explore here the most advanced functional assays across oncology indications, with an emphasis on tests already displaying a convincing clinical demonstration. We then recapitulate the main technical obstacles faced by researchers and clinicians to produce more accurate, and thus more predictive, models and the recent advances that have been developed to circumvent them. Finally, we summarize the regulatory and quality frameworks surrounding functional assays to ensure their safe and performant clinical implementation. Functional assays are valuable in vitro diagnostic tools that already stand beyond the “proof-of-concept” stage. Clinical studies show they have a major role to play by themselves but also in conjunction with molecular diagnostics. They now need a final lift to fully integrate the common armament used against cancers, and thus make their way into the clinical routine.
Keywords: Cancer, chemosensitivity, CSRA, functional assay, personalized medicine
 Global reach, higher impact
Global reach, higher impact