13.3
Impact Factor
Theranostics 2021; 11(19):9431-9451. doi:10.7150/thno.61902 This issue Cite
Research Paper
Interruption of neutrophil extracellular traps formation dictates host defense and tubular HOXA5 stability to augment efficacy of anti-Fn14 therapy against septic AKI
1. Department of Intensive Care Unit, Zhejiang Provincial People's Hospital, People's Hospital of Hangzhou Medical College, Hangzhou 310014, Zhejiang, P.R. China.
2. Zhejiang University School of Medicine, Zhejiang University, Hangzhou 310029, Zhejiang, P.R. China.
Abstract
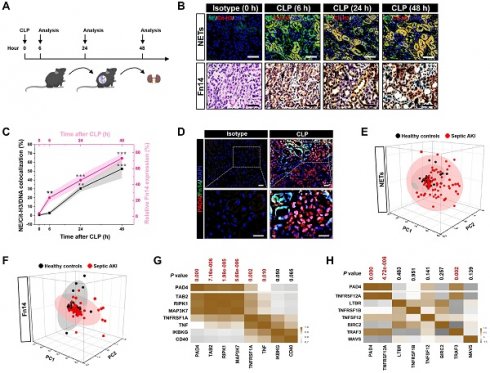
The immunosuppressive, inflammatory microenvironment orchestrated by neutrophil extracellular traps (NETs) plays a principal role in pathogenesis of sepsis. Fibroblast growth factor-inducible molecule 14 (Fn14) has been established as a potential target for septic acute kidney injury (AKI), making further therapeutic benefits from combined NETs and Fn14 blockade possible.
Methods: The concurrence of NETs and Fn14 in mice and patients with septic AKI were assessed by immunofluorescence, immunohistochemistry, enzyme-linked immunosorbent assay (ELISA) and in silico studies. Survival, histopathological and biochemical analyses of wild-type and PAD4-deficient CMV-Cre; PAD4fl/fl mice with septic AKI were applied to evaluate the efficacy of either pharmacological or genetic NETs interruption in combination with Fn14 blockade. Molecular mechanisms underlying such effects were determined by CRISPR technology, fluorescence-activated cell sorter analysis (FACS), cycloheximide (CHX) pulse-chase, luciferase reporter and chromatin immunoprecipitation (ChIP) assay.
Results: NETs formation is concurred with Fn14 upregulation in murine AKI models of abdominal, endotoxemic, multidrug-resistant sepsis as well as in serum samples of patients with septic AKI. Pharmacological or genetic interruption of NETs formation synergizes with ITEM-2, a monoclonal antibody (mAb) of Fn14, to prolong mice survival and provide renal protection against abdominal sepsis, the effects that could be abrogated by elimination of macrophages. Interrupting NETs formation predominantly perpetuates infiltration and survival of efferocytic growth arrest-specific protein 6+ (GAS6+) macrophages in combination with ITEM-2 therapy and enhances transcription of tubular cell-intrinsic Fn14 in a DNA methyltransferase 3a (DNMT3a)-independent manner through dismantling the proteasomes-mediated turnover of homeobox protein Hox-A5 (HOXA5) upon abdominal sepsis challenge or LPS stimuli. Pharmacological NETs interruption potentiates the anti-septic AKI efficacy of ITEM-2 in murine models of endotoxemic and multidrug-resistant sepsis.
Conclusion: Our preclinical data propose that interrupting NETs formation in combination with Fn14 mAb might be a feasible therapeutic strategy for septic AKI.
Keywords: Neutrophil extracellular traps, Fibroblast growth factor-inducible molecule 14, Macrophages, Homeobox protein Hox-A5, Septic acute kidney injury
 Global reach, higher impact
Global reach, higher impact