13.3
Impact Factor
Theranostics 2021; 11(19):9342-9357. doi:10.7150/thno.62255 This issue Cite
Research Paper
Neuromedin B receptor stimulation of Cav3.2 T-type Ca2+ channels in primary sensory neurons mediates peripheral pain hypersensitivity
1. Department of Geriatrics & Institute of Neuroscience, the Second Affiliated Hospital of Soochow University, Suzhou 215004, China.
2. Comprehensive Pneumology Center, Helmholtz Zentrum München, Munich 81377, Germany.
3. Department of Physiology and Neurobiology & Centre for Ion Channelopathy, Medical College of Soochow University, Suzhou 215123, China.
4. Department of Endocrinology, Shanghai East Hospital, Tongji University School of Medicine, Shanghai 200120, China.
5. Jiangsu Key Laboratory of Neuropsychiatric Diseases, Soochow University, Suzhou 215123, China.
#These authors contributed equally to this work.
Abstract
Background: Neuromedin B (Nmb) is implicated in the regulation of nociception of sensory neurons. However, the underlying cellular and molecular mechanisms remain unknown.
Methods: Using patch clamp recording, western blot analysis, immunofluorescent labelling, enzyme-linked immunosorbent assays, adenovirus-mediated shRNA knockdown and animal behaviour tests, we studied the effects of Nmb on the sensory neuronal excitability and peripheral pain sensitivity mediated by Cav3.2 T-type channels.
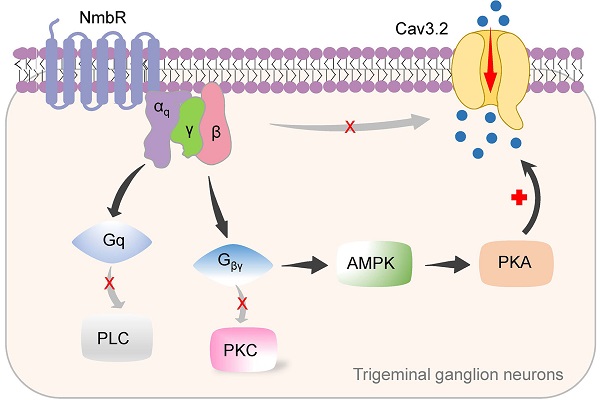
Results: Nmb reversibly and concentration-dependently increased T-type channel currents (IT) in small-sized trigeminal ganglion (TG) neurons through the activation of neuromedin B receptor (NmbR). This NmbR-mediated IT response was Gq protein-coupled, but independent of protein kinase C activity. Either intracellular application of the QEHA peptide or shRNA-mediated knockdown of Gβ abolished the NmbR-induced IT response. Inhibition of protein kinase A (PKA) or AMP-activated protein kinase (AMPK) completely abolished the Nmb-induced IT response. Analysis of phospho-AMPK (p-AMPK) revealed that Nmb significantly activated AMPK, while AMPK inhibition prevented the Nmb-induced increase in PKA activity. In a heterologous expression system, activation of NmbR significantly enhanced the Cav3.2 channel currents, while the Cav3.1 and Cav3.3 channel currents remained unaffected. Nmb induced TG neuronal hyperexcitability and concomitantly induced mechanical and thermal hypersensitivity, both of which were attenuated by T-type channel blockade. Moreover, blockade of NmbR signalling prevented mechanical hypersensitivity in a mouse model of complete Freund's adjuvant-induced inflammatory pain, and this effect was attenuated by siRNA knockdown of Cav3.2.
Conclusions: Our study reveals a novel mechanism by which NmbR stimulates Cav3.2 channels through a Gβγ-dependent AMPK/PKA pathway. In mouse models, this mechanism appears to drive the hyperexcitability of TG neurons and induce pain hypersensitivity.
Keywords: T-type Ca2+ channel, neuromedin B receptor, trigeminal ganglion neurons, protein kinase A
 Global reach, higher impact
Global reach, higher impact